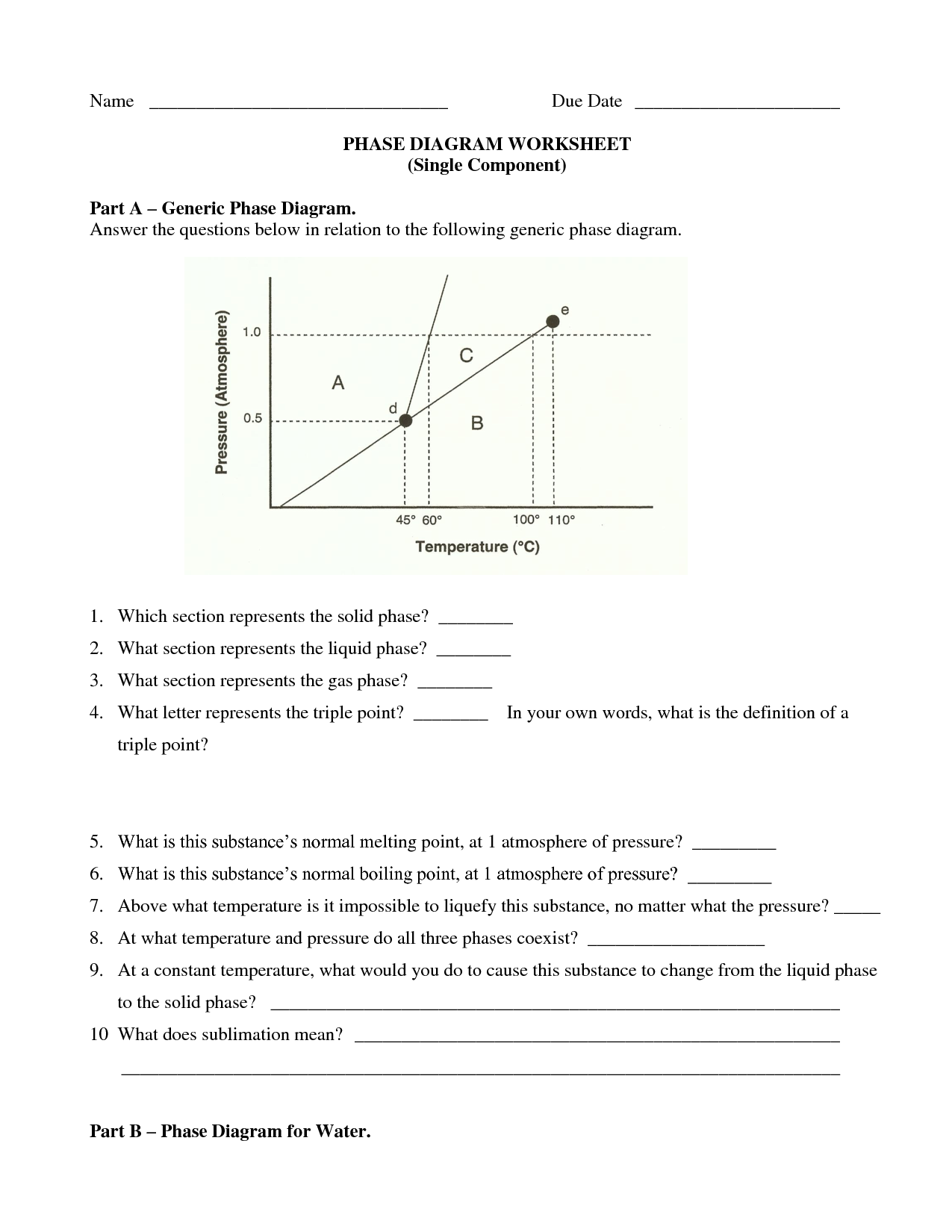
Phase Change Diagram Worksheet
If you're teaching a science class or simply want to dive deeper into understanding the various phases of matter and their transitions, then a phase change diagram worksheet could be just what you need.
Table of Images 👆
- Energy Phase Change Diagram
- Label Phase Change Diagram
- Phase Change Worksheet Answer Sheet
- Phase Diagram Worksheet Answer Key
- Phase Change Worksheet Answer Key
- Phase Change Diagram Worksheet Answers
- Chemistry Phase Diagram Worksheet
- Phase Change Worksheet Answers
- Blank Phase Change Diagram
- States of Matter Phase Changes Worksheet
More Other Worksheets
Kindergarten Worksheet My RoomSpanish Verb Worksheets
Cooking Vocabulary Worksheet
DNA Code Worksheet
Meiosis Worksheet Answer Key
Art Handouts and Worksheets
7 Elements of Art Worksheets
All Amendment Worksheet
Symmetry Art Worksheets
Daily Meal Planning Worksheet
What is a phase change diagram?
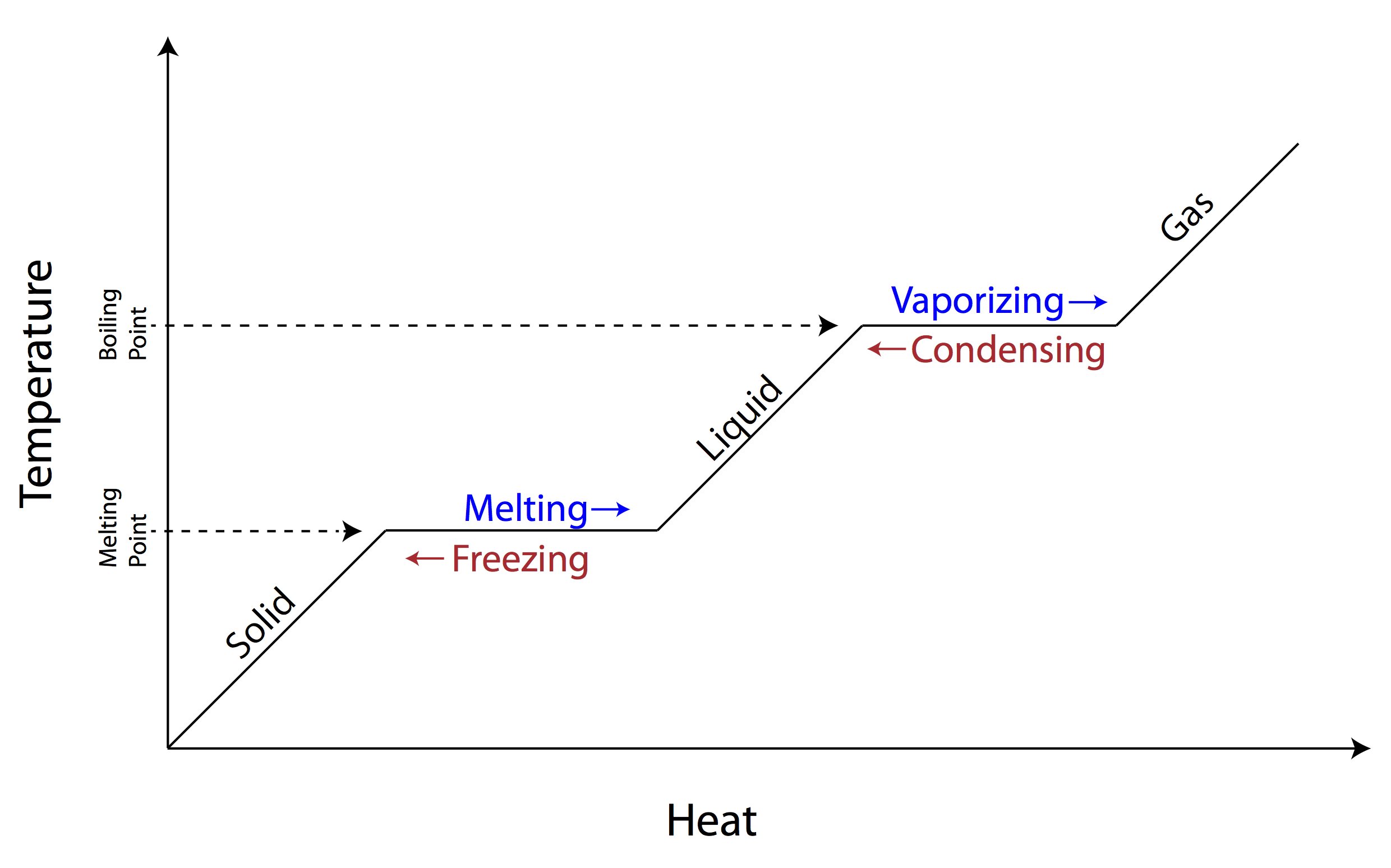
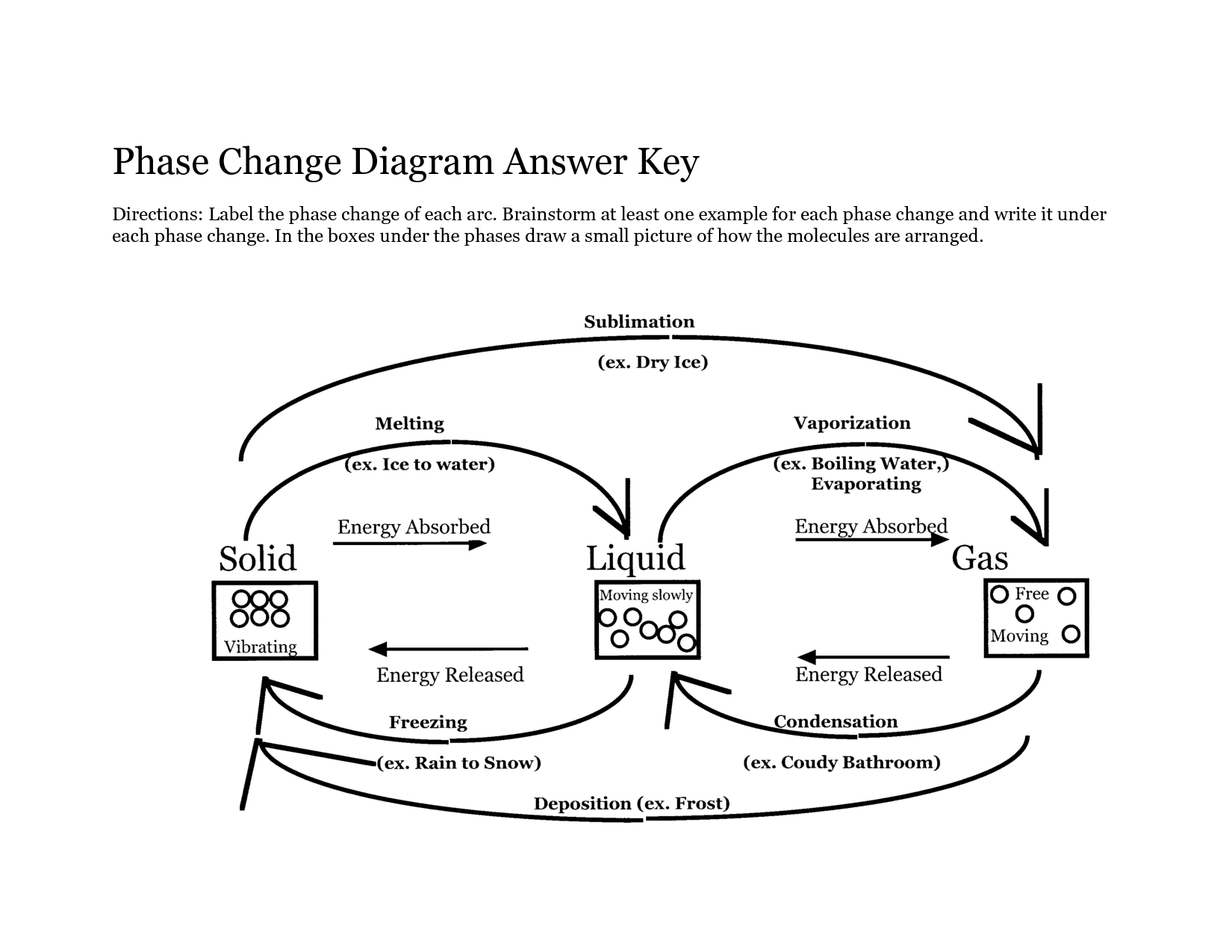
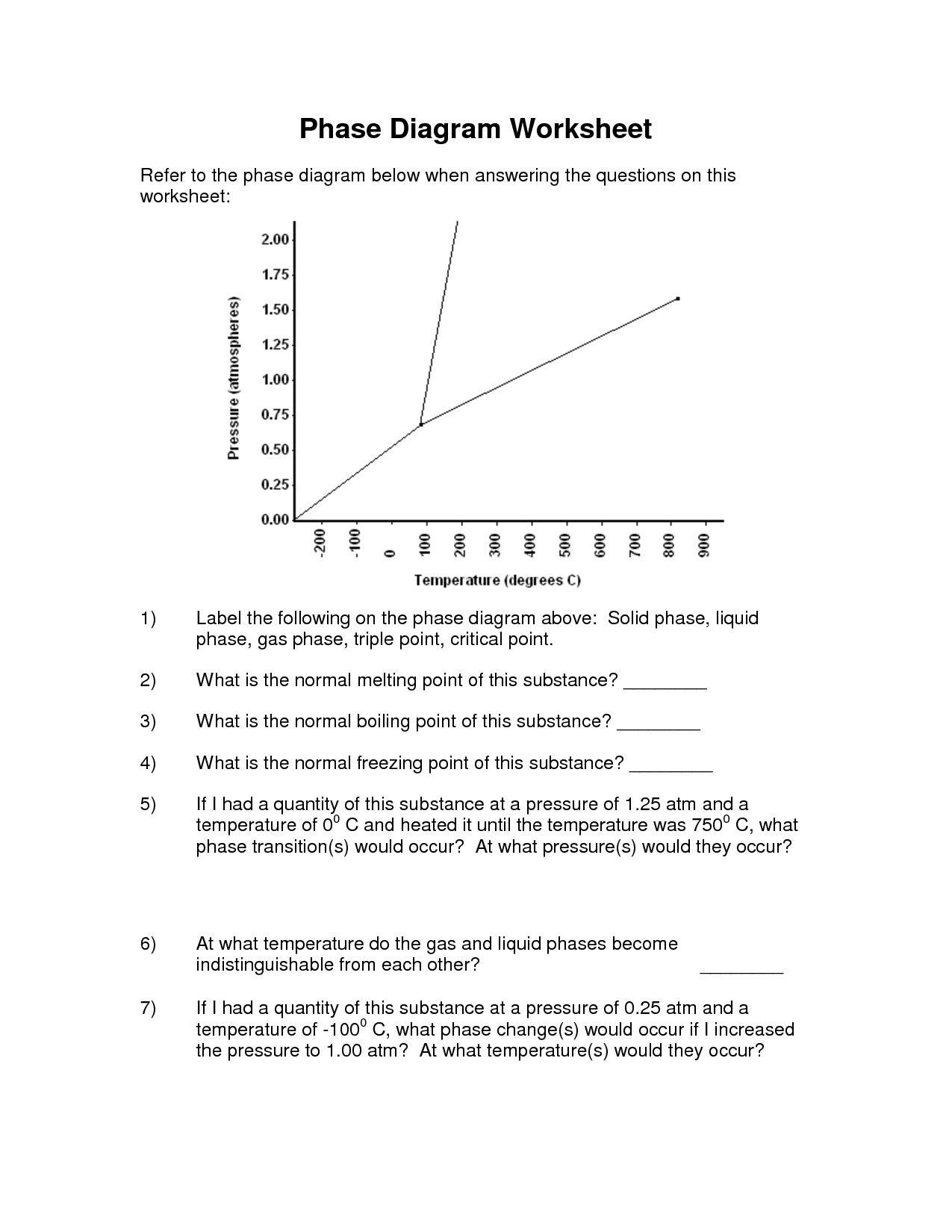
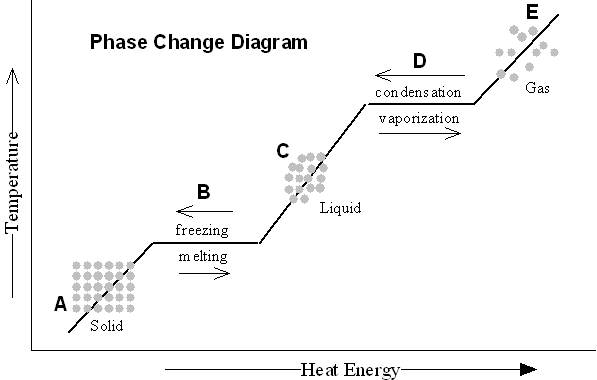
A phase change diagram is a graphical representation that illustrates how the physical state of a substance changes as a function of temperature and pressure. It typically shows the equilibrium between solid, liquid, and gas phases of a substance, depicting the boundaries where phase transitions occur such as melting, freezing, evaporation, and condensation. The diagram helps to visualize the conditions under which a substance transitions between different phases.
What are the three main phases of matter?
The three main phases of matter are solid, liquid, and gas. Solids have a definite shape and volume, liquids have a definite volume but take the shape of their container, and gases have neither a definite shape nor volume and fill the space available to them.
What does the x-axis represent on a phase change diagram?
The x-axis on a phase change diagram represents the amount of heat added or removed from a substance, typically measured in units of energy such as joules or calories. It shows how the temperature of the substance changes as heat is transferred during phase transitions, allowing us to visualize the energy required for the substance to change from one phase to another.
What does the y-axis represent on a phase change diagram?
The y-axis on a phase change diagram represents the temperature of a substance. It shows the temperature at which phase changes occur for a substance under different conditions of pressure or heat.
What is the melting point of a substance?
The melting point of a substance is the temperature at which it changes from a solid to a liquid phase. It is a specific characteristic property of the substance and is typically measured in degrees Celsius or Fahrenheit.
What is the boiling point of a substance?
The boiling point of a substance is the temperature at which it changes state from liquid to gas at a constant pressure. It is the temperature at which the vapor pressure of the liquid equals the ambient pressure, leading to the formation of bubbles of vapor within the liquid.
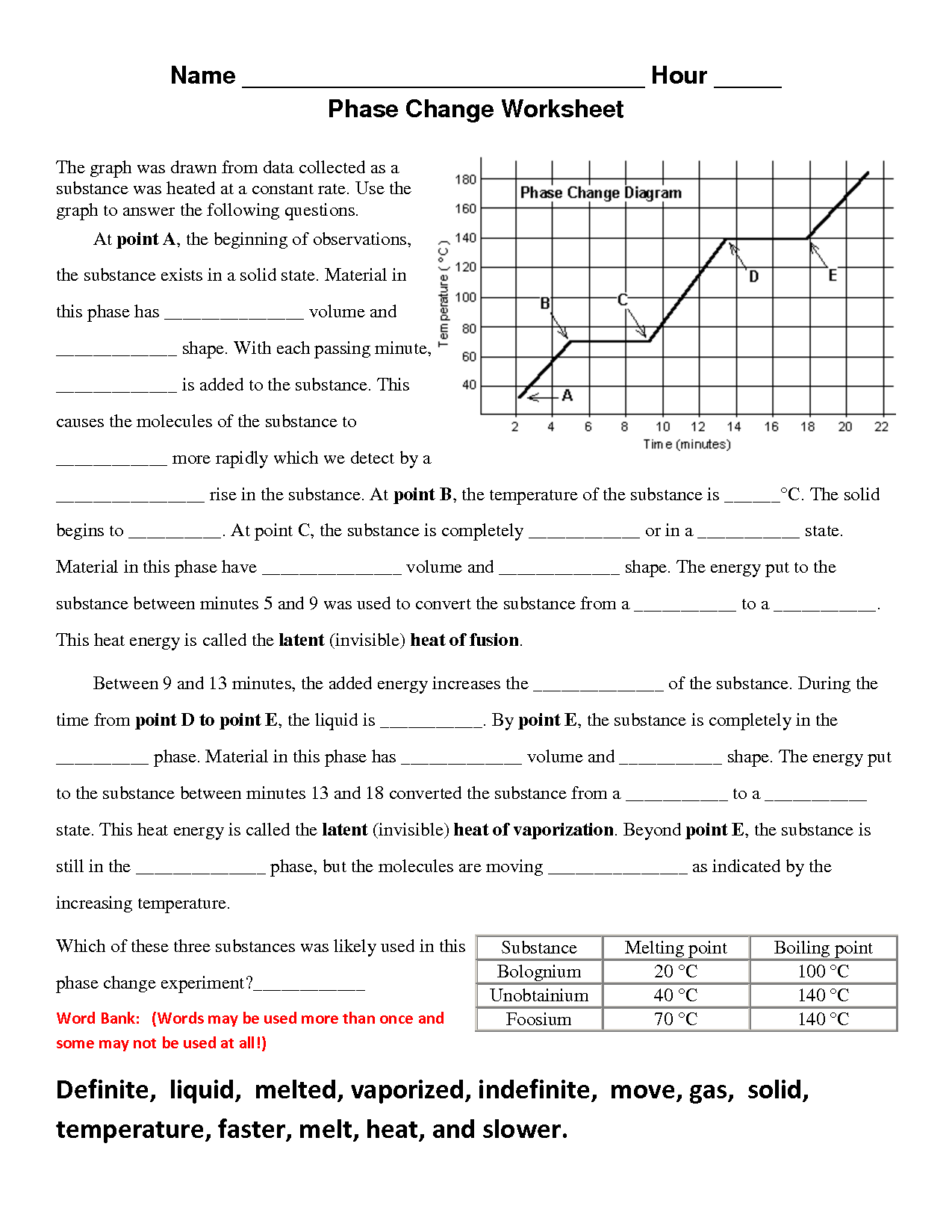
What happens to the temperature of a substance during a phase change?
During a phase change, the temperature of a substance remains constant as the heat energy is being used to break the intermolecular forces holding the particles together rather than increasing the kinetic energy of the particles. This energy is known as latent heat and is absorbed or released during a phase change, causing a plateau in the temperature until the phase change is complete.
What is the process called when a substance changes from a solid to a liquid?
The process of a substance changing from a solid to a liquid is called melting.
What is the process called when a substance changes from a liquid to a gas?
The process is called vaporization, specifically when a substance changes from a liquid to a gas.
What is the process called when a substance changes directly from a solid to a gas?
The process is called sublimation.
Have something to share?
Who is Worksheeto?
At Worksheeto, we are committed to delivering an extensive and varied portfolio of superior quality worksheets, designed to address the educational demands of students, educators, and parents.






















Comments