Physical Changes Matter Worksheets
Physical changes matter worksheets are an excellent resource for educators and parents who are seeking to reinforce the understanding of this scientific concept among their students and children. Designed to provide a comprehensive overview of physical changes, these worksheets cover a range of topics and activities that engage learners in exploring the properties of matter and how they can be altered.
Table of Images 👆
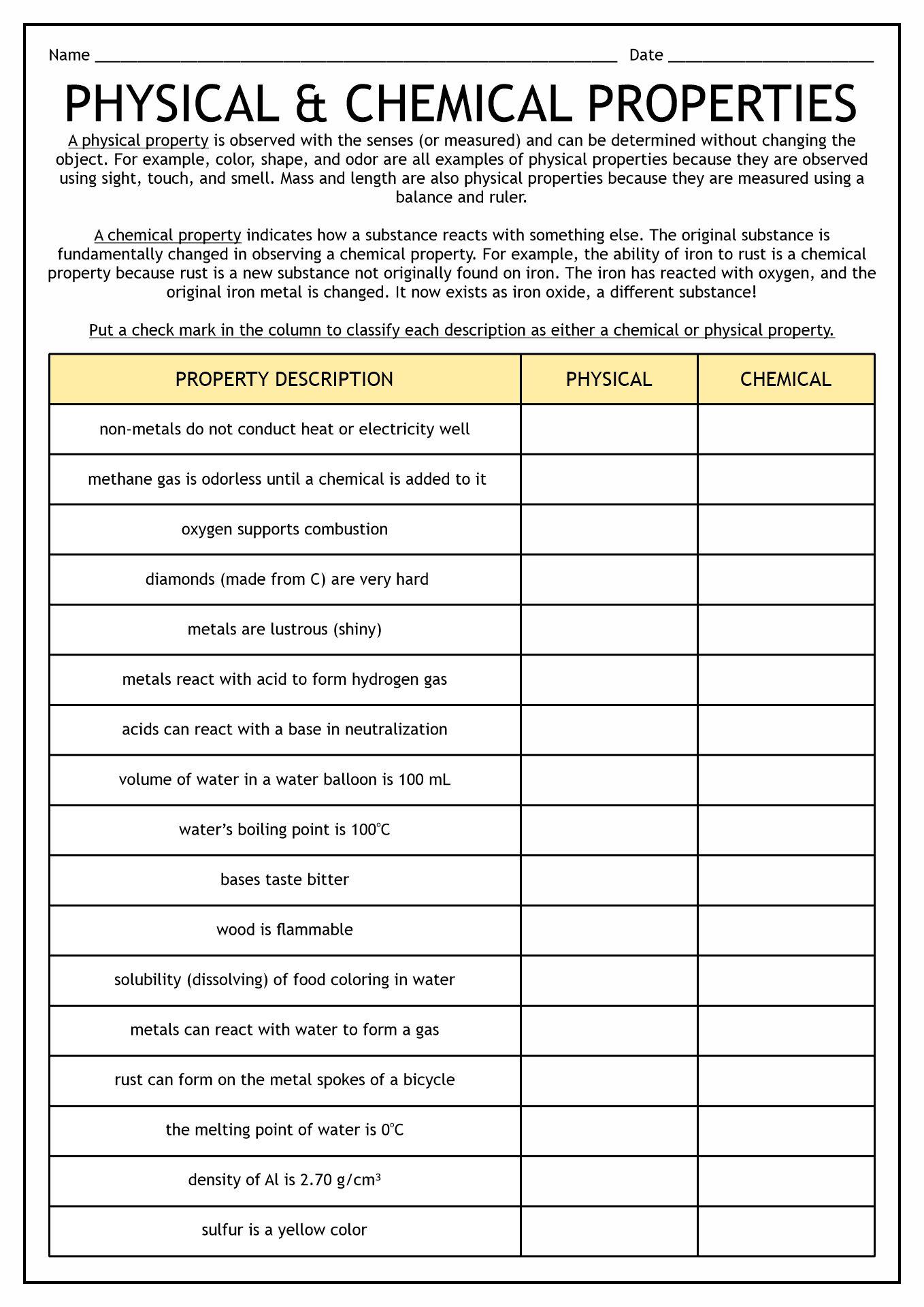
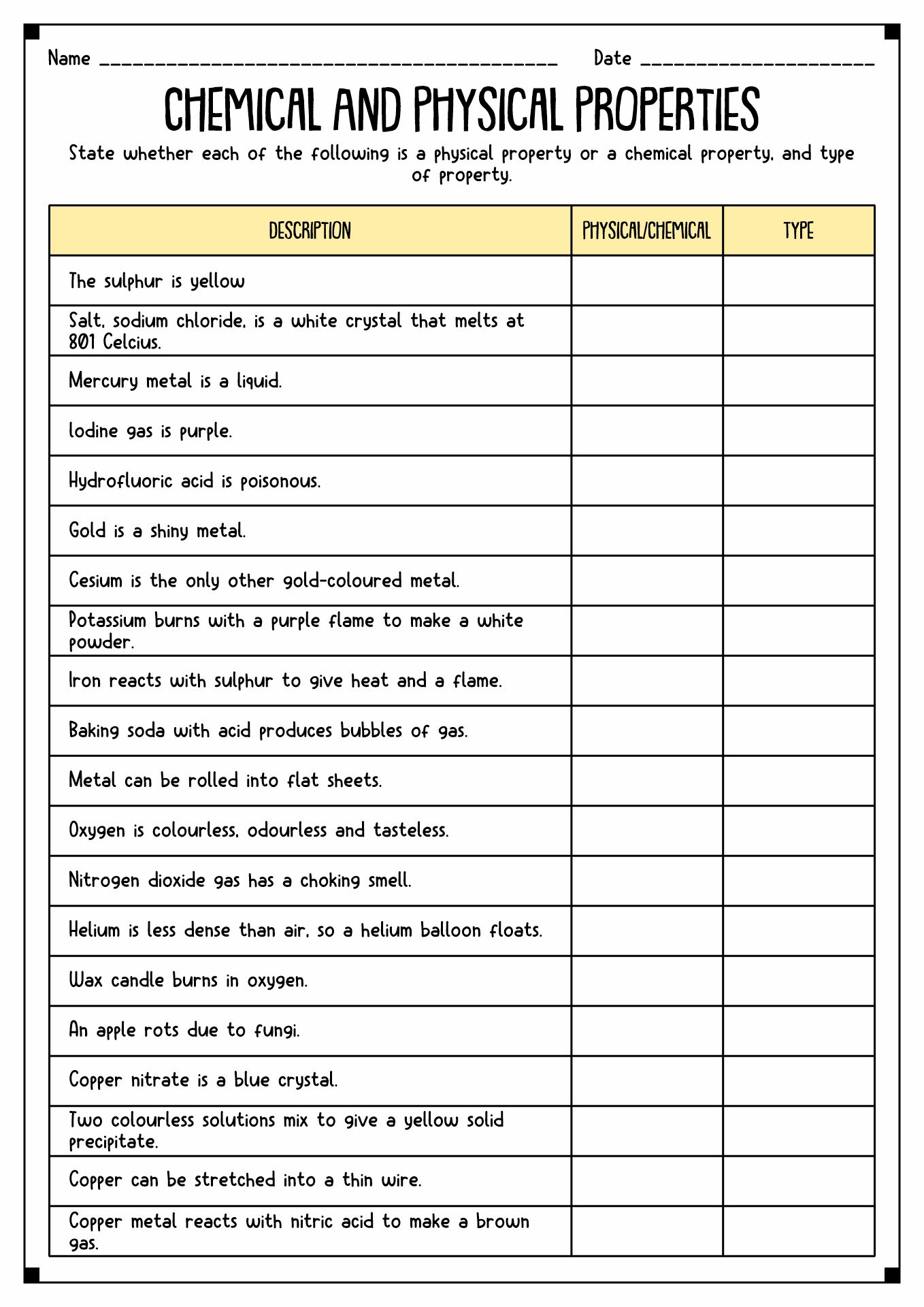
- Physical vs Chemical Properties Worksheet
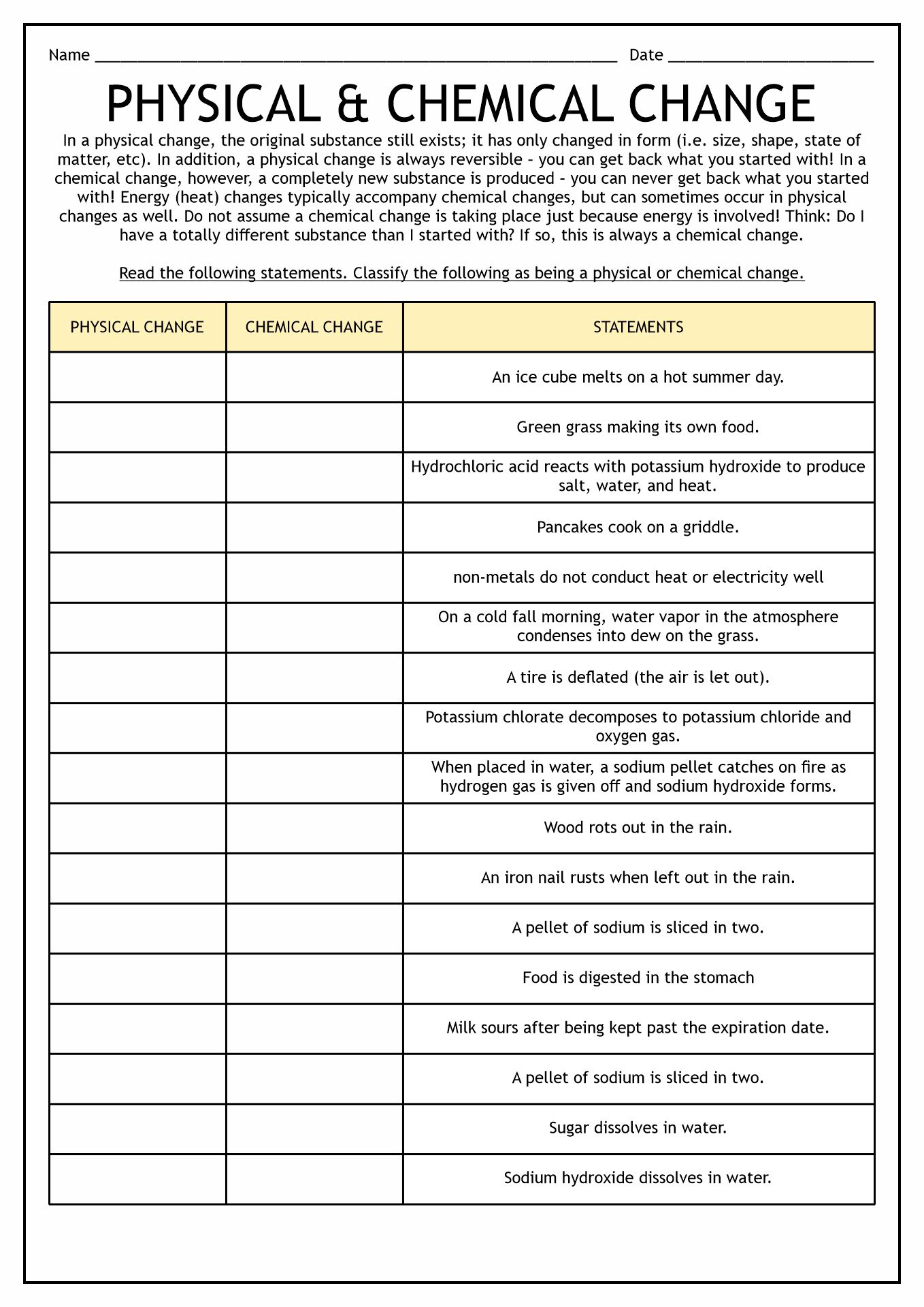
- Matter Physical and Chemical Changes Worksheet
- Kids Physical and Chemical Changes Worksheet
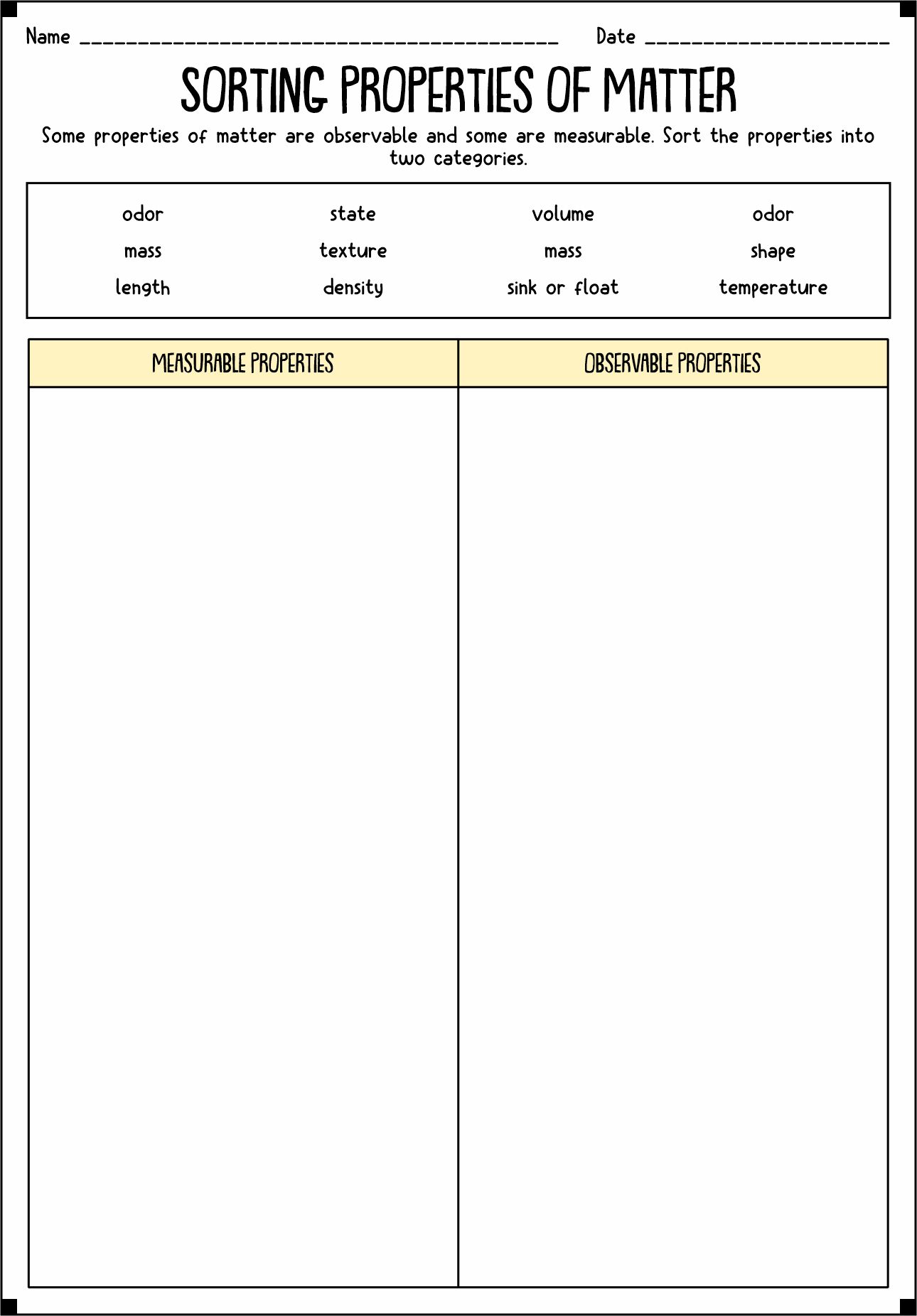
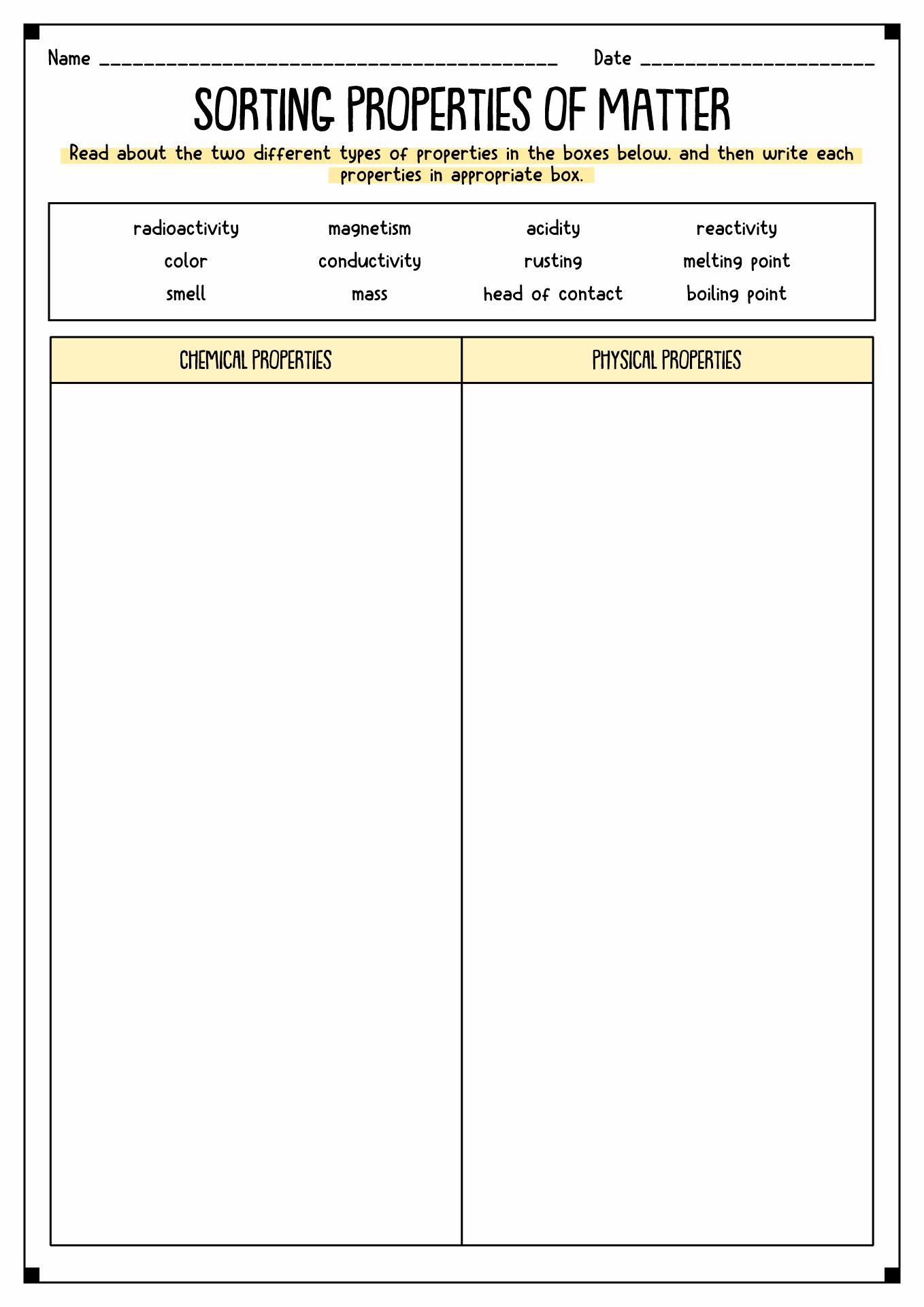
- Properties of Matter Sort
- Physical Properties of Matter Worksheet
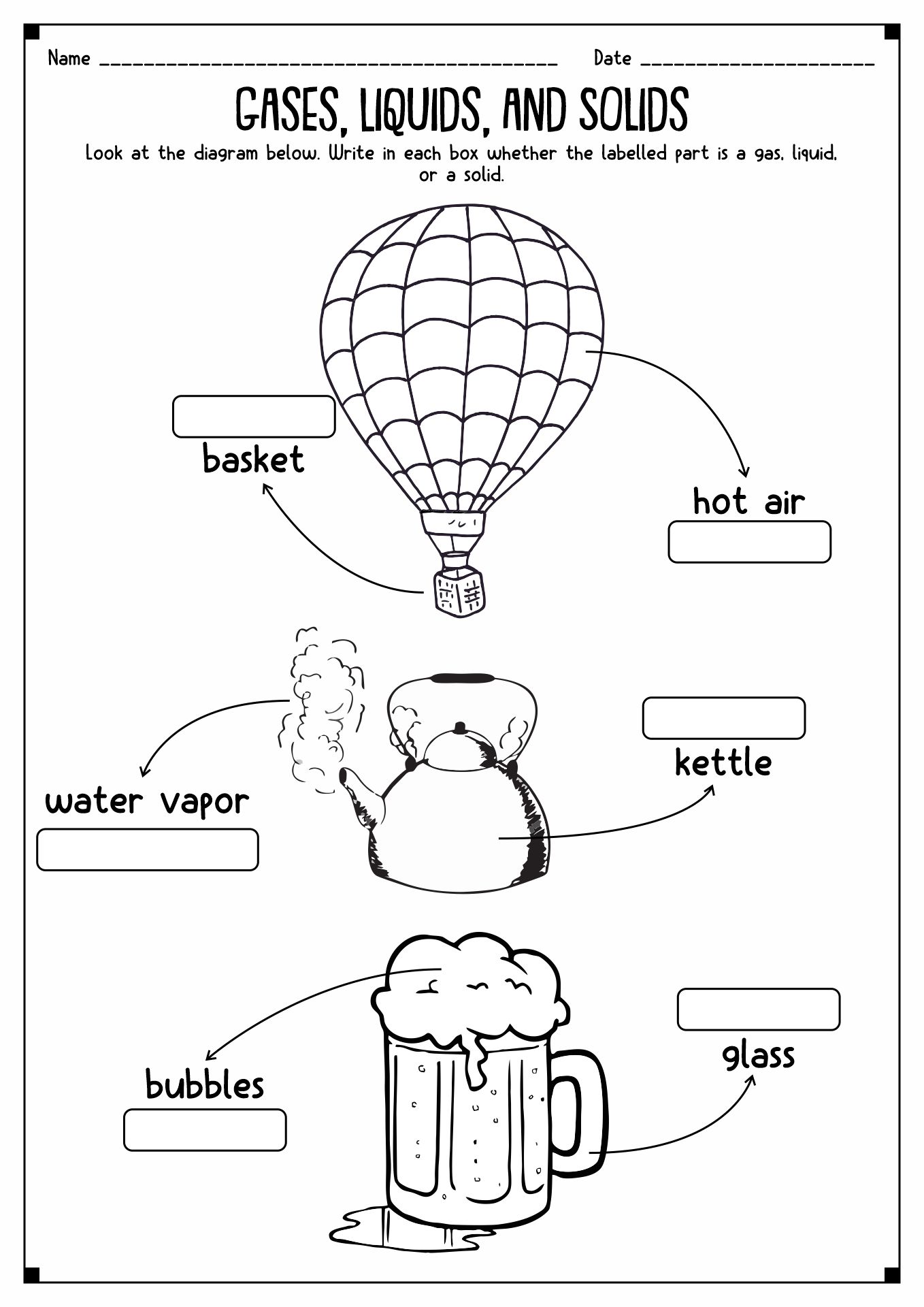
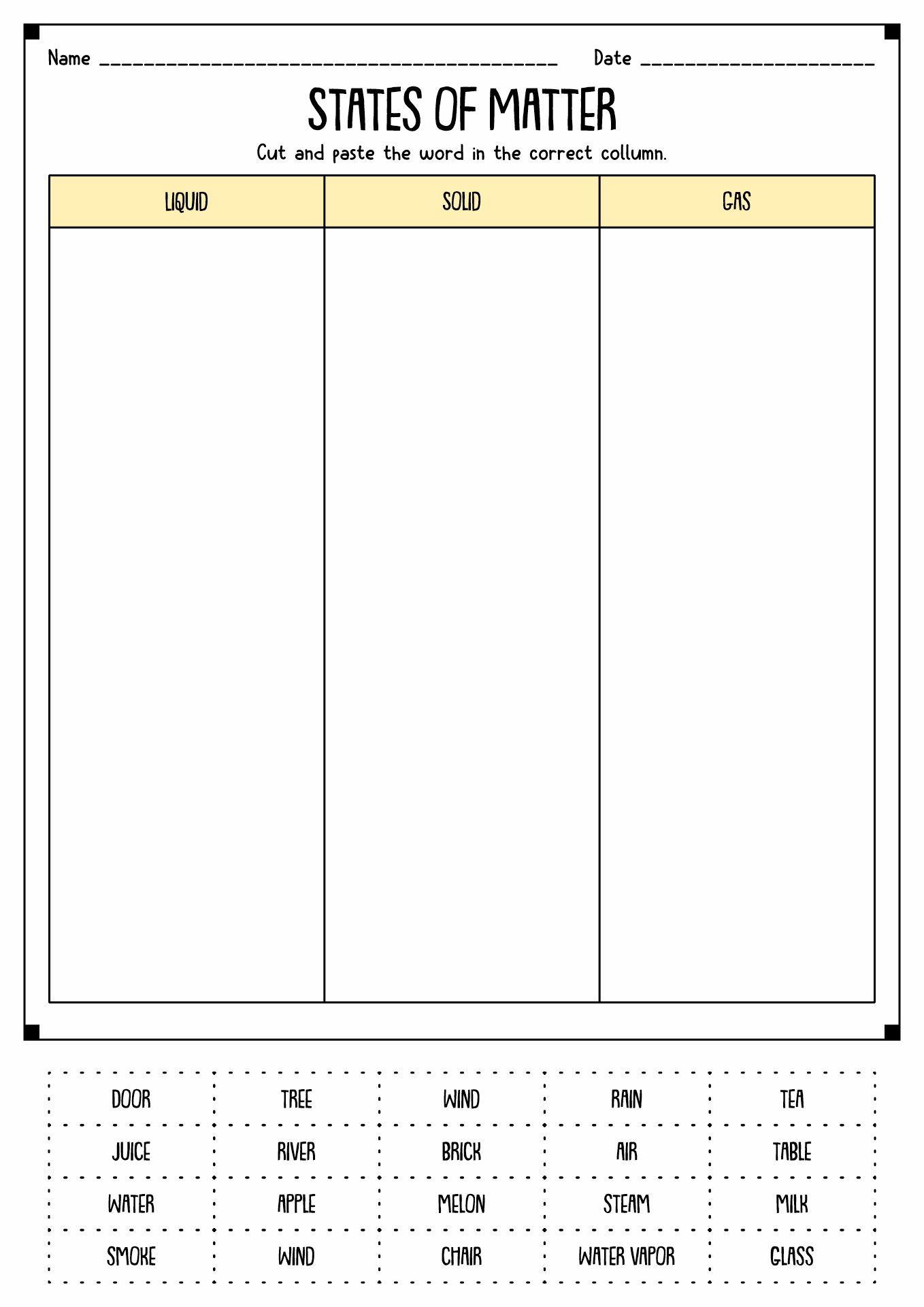
- Solid-Liquid Gas Worksheet
- Physical Properties of Matter Worksheets for Kids
- Physical Science Glencoe Note Taking Worksheet Answer Key
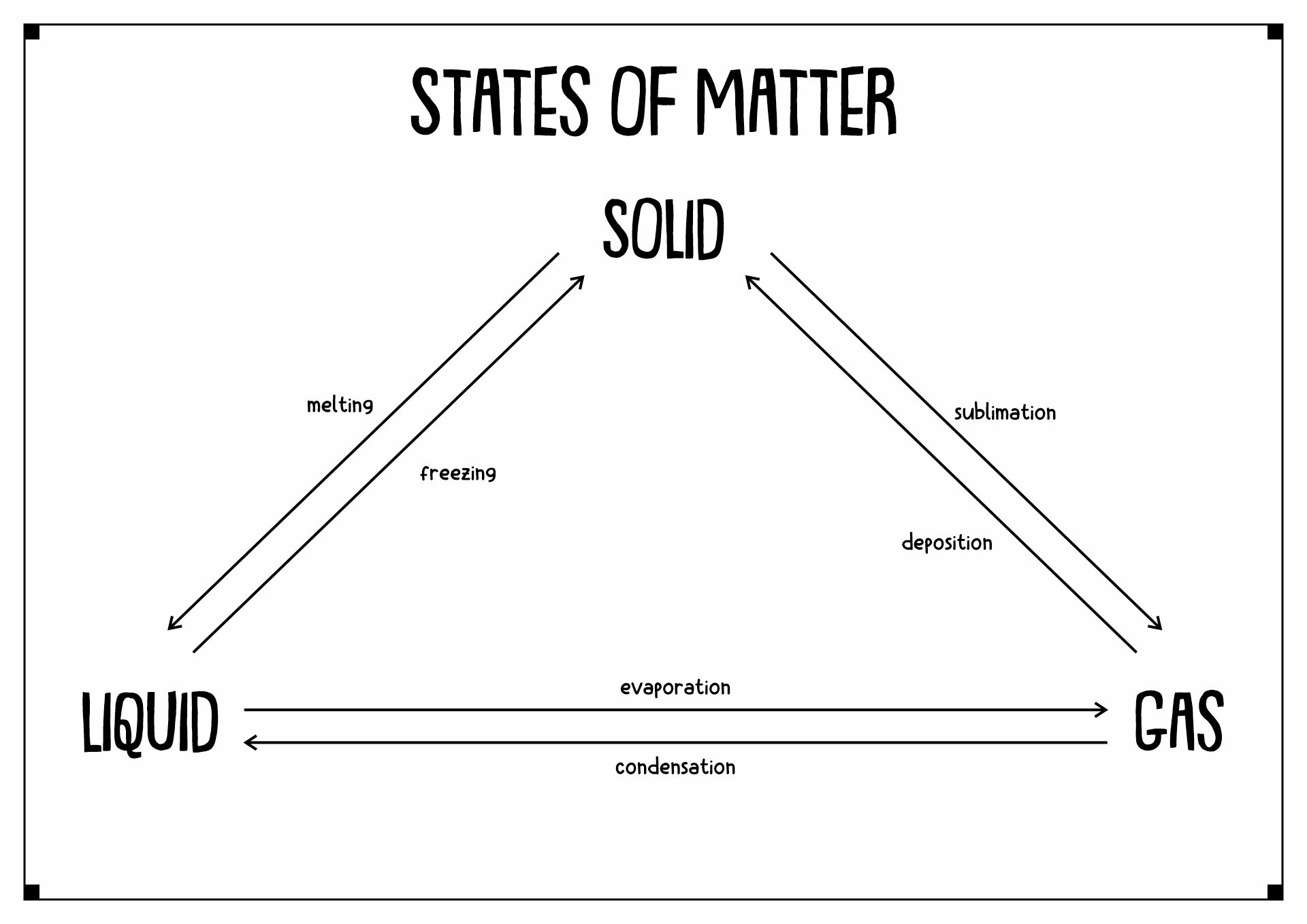
- States of Matter Solid-Liquid Gas
- Physical Properties and Chemical Changes Worksheet
- States of Matter Worksheets 2nd Grade
- Chemistry Elements and Symbols Worksheets
- Physical vs Chemical Change Worksheet
- Science Properties of Matter Worksheets
- Science Properties of Matter Worksheets
- Science Properties of Matter Worksheets

Understanding the physical changes in matter is crucial, and with our Physical Changes Matter Worksheets, you can enhance your educational resources effectively.
More Other Worksheets
Kindergarten Worksheet My RoomSpanish Verb Worksheets
Spring Clothes Worksheet
For First Grade Phonics Worksheets
Hundreds Chart Missing Numbers Worksheet
Healthy Eating Plate Printable Worksheet
Cooking Vocabulary Worksheet
My Shadow Worksheet
Large Printable Blank Pyramid Worksheet
Relationship Circles Worksheet
Congeal your science with these Physical Changes Matter Worksheets!
Summary: There are two ways of matter changes, physical and chemical. In a physical change, the substance of the physical properties will change. Alteration in form, color, odor, and solubility are examples of physical change. Learning about water will give us a real-life example of this transformation. Many professionals agree that learning about matter will be a concrete foundation for young students to learn advanced chemistry and physics. It also opens the door for them to understand their environment.
What is Physical Change?
Everything in this world is built from matter. According to Oxford Dictionaries, it refers to the physical substance which fills the space and possesses rest masses. In a more simple definition, it is the foundation of every object in the world. Humans cannot make or destroy them. However, through natural circumstances, matter can change. It cycles through the world without stopping. They include molecules, atoms, particles, and substances.
There are two ways of matter changes, physical and chemical. The matter's amount stays the same before and after the change. This phenomenon is what the scientist called as Law of Conservation Mass. In a physical change, the substance of the physical properties will change. Alteration in form, color, odor, and solubility are examples of physical change. Learning about water will give us a real-life example of this transformation. Water is an element that can exist in three states, solid, liquid, and gas. It should experience a physical change to reform from one state to another. However, there is no change in the amount during the process. We can reverse the results in physical change.
What Does it Mean by Chemical Change?
As we learn from the previous part, there are two ways of matter change, physical and chemical. In this part, we will discuss the chemical changes. The distinct difference between these two alterations is that the first changes did not create new substances; chemical changes do. We cannot reverse the result of the latter. In conclusion, chemical changes produce unreversible new substances.
A chemical change occurs when there is a modification of the matter's composition. It includes some processes, such as forming and breaking chemical bonds during the chemical reaction. This process affects the arrangement and creates a new substance from the chemical reaction. This creation is almost impossible to be reverted to its original state. In many cases, it is formidable to spot chemical changes. The experts stated we can examine if the object has a color, odor, temperature, precipitate appearance, and gas formation to help us identify it. Rotting food, rusting on iron, combustion, and fermentation are chemical changes in our daily life.
How Many Properties of Matters Are There?
The matter has properties. It refers to the traits of the object that we can measure. There are two types of properties of matter, physical and chemical. Physical Properties refer to the characteristics we can calculate without changing the arrangements. Mass, color, and volume are some examples of Physical Properties. Meanwhile, Chemical Properties are the ability of a substance to react to create a new essence.
Physical Properties have two types, intensive and extensive. Intensive properties mean a system of independent local physical elements. Color, melting and boiling point, and density are examples of Intensive Properties. Meanwhile, Extensive Properties are dependent on the amount of the matter sample. Mass, volume, length, and shape are instances of Extensive Properties.
There are four Chemicals Properties that the students should master:
- Reactivity: It is the preference of a substance to mix with others. Some elements are reactive. Meanwhile, the others are inactive.
- Flammability: Flammability is the traits of an element to get burned. When it burns, it reacts to oxygen and changes into other substances.
- Toxicity: It refers to the level of how a substance can harm an organism or the environment.
- Acidity: Acidity means how a substance reacts with acid elements.
How to Explain States of Matter?
States of matter is how a substance can take form in the universe. There are four kinds of them, gas, solid, liquid, and plasma. Solid is a stage of an object when the particles inside it are packed and secured together with almost no motion. It has a concrete shape with concrete mass and volume. In liquid states, the particles inside the material move more loosely and can hover around each other. These movements cause the matter not to have a concrete shape. Gas substances have spacious space around the particles with high kinetic energy. Last but not least is plasma. It is a gas with electricity. Sun and stars are examples of matter with plasma.
Why Should We Learn About Matter?
Learning about the matter is unavoidable when we study science. It has various benefits for our daily life application. It also has a vital role in world development. Many professionals agree that learning about matter will be a concrete foundation for young students to learn advanced chemistry and physics. It also opens the door for them to understand their environment. Learning about matter also helps us to appreciate and respect our surroundings, as everything consists of them.
Have something to share?
Popular Categories
Who is Worksheeto?
At Worksheeto, we are committed to delivering an extensive and varied portfolio of superior quality worksheets, designed to address the educational demands of students, educators, and parents.


























Comments
These Physical Changes Matter Worksheets are a helpful and engaging resource for teaching students about physical changes. They provide a great opportunity for hands-on learning and critical thinking. Highly recommended!
Great resource for teaching physical changes! The worksheets are clear and provide a practical approach to understanding the topic. Highly recommended for engaging and interactive lessons.
These Physical Changes Matter Worksheets are a valuable resource for teaching students about the different types of physical changes. The clear and concise design makes it easy to understand and engage with the material. A great tool to enhance learning!
Very helpful worksheets! They allow my students to easily grasp and understand the concept of physical changes. Thank you for providing this printable resource!
I found the Physical Changes Matter Worksheets very helpful in explaining different physical changes in a simple and concise manner. It made learning about the topic enjoyable and easy to understand. Thank you for providing such a valuable resource!