Matter Worksheets for High School
Are you a high school student or teacher looking for engaging and thought-provoking worksheets on matter? Well, search no more! We have just what you need to enhance your understanding of this fascinating subject. Our matter worksheets are designed to provide comprehensive review and practice, covering various topics related to the properties, states, and changes of matter. Whether you're just starting out or need some extra practice, our worksheets will help you sharpen your knowledge and excel in your studies of matter.
Table of Images 👆
More Other Worksheets
Kindergarten Worksheet My RoomSpanish Verb Worksheets
Cooking Vocabulary Worksheet
DNA Code Worksheet
Meiosis Worksheet Answer Key
Art Handouts and Worksheets
7 Elements of Art Worksheets
All Amendment Worksheet
Symmetry Art Worksheets
Daily Meal Planning Worksheet
What is matter?
Matter is anything that has mass and occupies space. It is made up of atoms, which are the building blocks of all substances in the universe. Matter can exist in various forms such as solid, liquid, and gas, and it can undergo physical and chemical changes.
What are the three states of matter?
The three states of matter are solid, liquid, and gas.
How does matter change from one state to another?
Matter changes from one state to another through either increasing or decreasing temperature or pressure. When heat is added to a solid, its particles gain energy and begin to move more, eventually breaking the bonds that hold them together in a fixed position, leading to a change from a solid to a liquid state (melting). Further heating will cause the particles to move even more freely, turning the liquid into a gas (evaporation). On the other hand, decreasing temperature or increasing pressure can cause a gas to condense into a liquid (condensation) and solidify into a solid (freezing). These phase changes involve the rearrangement of particles and changes in their energy levels, resulting in the transition of matter from one state to another.
What is the difference between physical and chemical properties of matter?
Physical properties of matter are characteristics that can be observed or measured without changing the composition of the material, such as color, density, and melting point. On the other hand, chemical properties involve the behavior of a substance in reactions that lead to the formation of new substances, like flammability, reactivity, and acidity. Chemical properties describe how a substance interacts with other substances, whereas physical properties describe the characteristics of a substance without changing its chemical identity.
What are elements and compounds?
Elements are substances made up of only one type of atom, which cannot be broken down into simpler substances through chemical reactions. Compounds, on the other hand, are substances composed of two or more different types of atoms chemically bonded together in fixed ratios. While elements retain their properties, compounds have unique properties different from the elements they are made of.
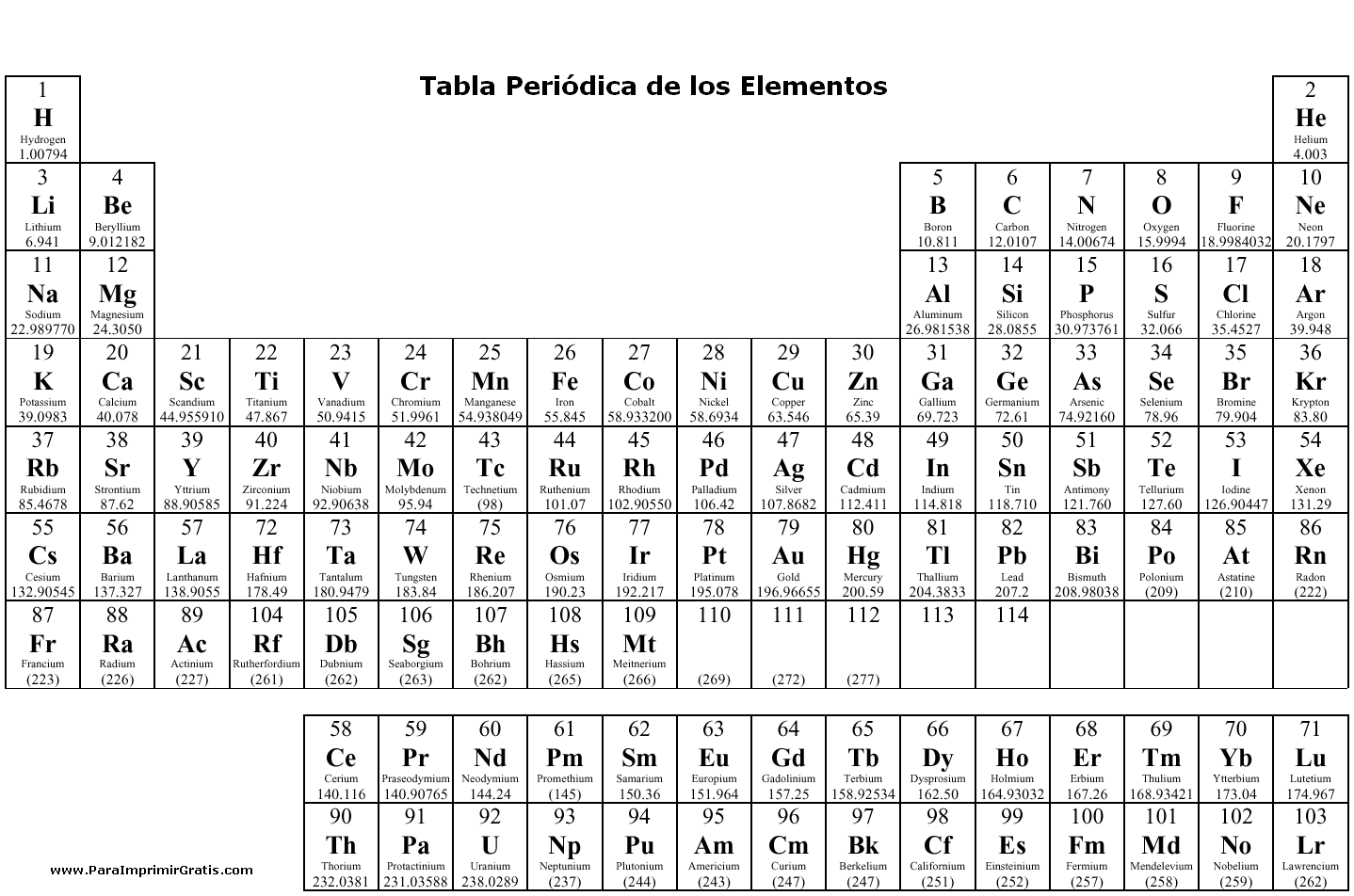
How are elements represented in the periodic table?
Elements in the periodic table are represented by their chemical symbol, which is a one- or two-letter abbreviation derived from their name. Each element is also assigned a unique atomic number based on the number of protons in its nucleus, which determines its position in the periodic table. Additionally, elements are organized in the periodic table based on their atomic number and similar chemical properties, with rows called periods and columns called groups or families.
What are the basic building blocks of matter?
The basic building blocks of matter are atoms, which are made up of protons, neutrons, and electrons. Protons and neutrons are located in the nucleus of an atom, while electrons orbit the nucleus. These particles combine in different ways to form elements, which are the fundamental substances that make up all matter in the universe.
What is the law of conservation of mass?
The law of conservation of mass states that in a closed system, mass is neither created nor destroyed in a chemical reaction or a physical transformation. This means that the total mass of the substances before a reaction is equal to the total mass of the substances after the reaction. Mass can be rearranged, combined, or separated, but it remains constant.
How do you calculate the density of a substance?
To calculate the density of a substance, you need to divide the mass of the substance by its volume. The formula for density is: Density = Mass / Volume. Mass is typically measured in grams or kilograms, while volume is measured in cubic centimeters or cubic meters. By dividing the mass by the volume, you can determine how tightly packed the particles of the substance are, providing a measure of its density.
What are some examples of physical and chemical changes in matter?
Some examples of physical changes in matter include melting ice, boiling water, and dissolving sugar in water. Chemical changes, on the other hand, involve the formation of new substances such as rusting of iron, baking a cake, and burning wood. These changes can be identified by observing differences in the properties of the substances before and after the change occurs.
Have something to share?
Who is Worksheeto?
At Worksheeto, we are committed to delivering an extensive and varied portfolio of superior quality worksheets, designed to address the educational demands of students, educators, and parents.























Comments