Chemistry Conversion Factors Worksheet
Teaching chemistry often feels like balancing two worlds at once: numbers and concepts. Many students understand the idea behind a lesson but get stuck when math enters the picture, especially when unit conversions are involved. This is where a Chemistry Conversion Factors Worksheet becomes a powerful support tool for both teachers and parents!
Instead of asking students to jump straight into complex calculations, this type of worksheet slows the process down in a helpful way. Learners see how numbers, units, and ratios connect step by step, which reduces confusion and careless mistakes.
So, whether you’re working in a classroom, tutoring one-on-one, or supporting learning at home, these worksheets help turn abstract conversions into something visible, logical, and far less intimidating!
Table of Images 👆
- Metric Conversion Table Chart
- Dimensional Analysis Conversion Chart
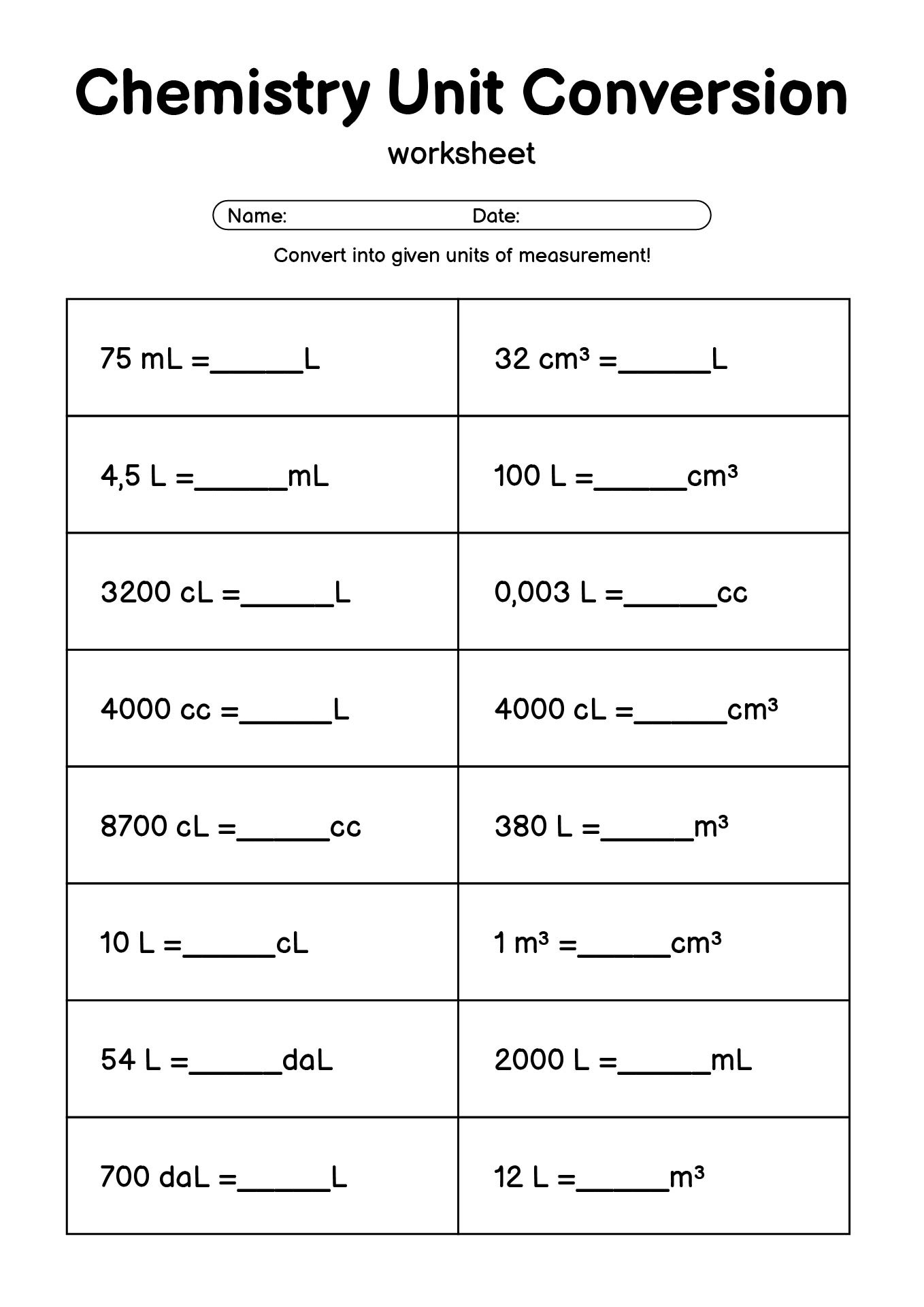
- Chemistry Conversion Factors Lesson Worksheet
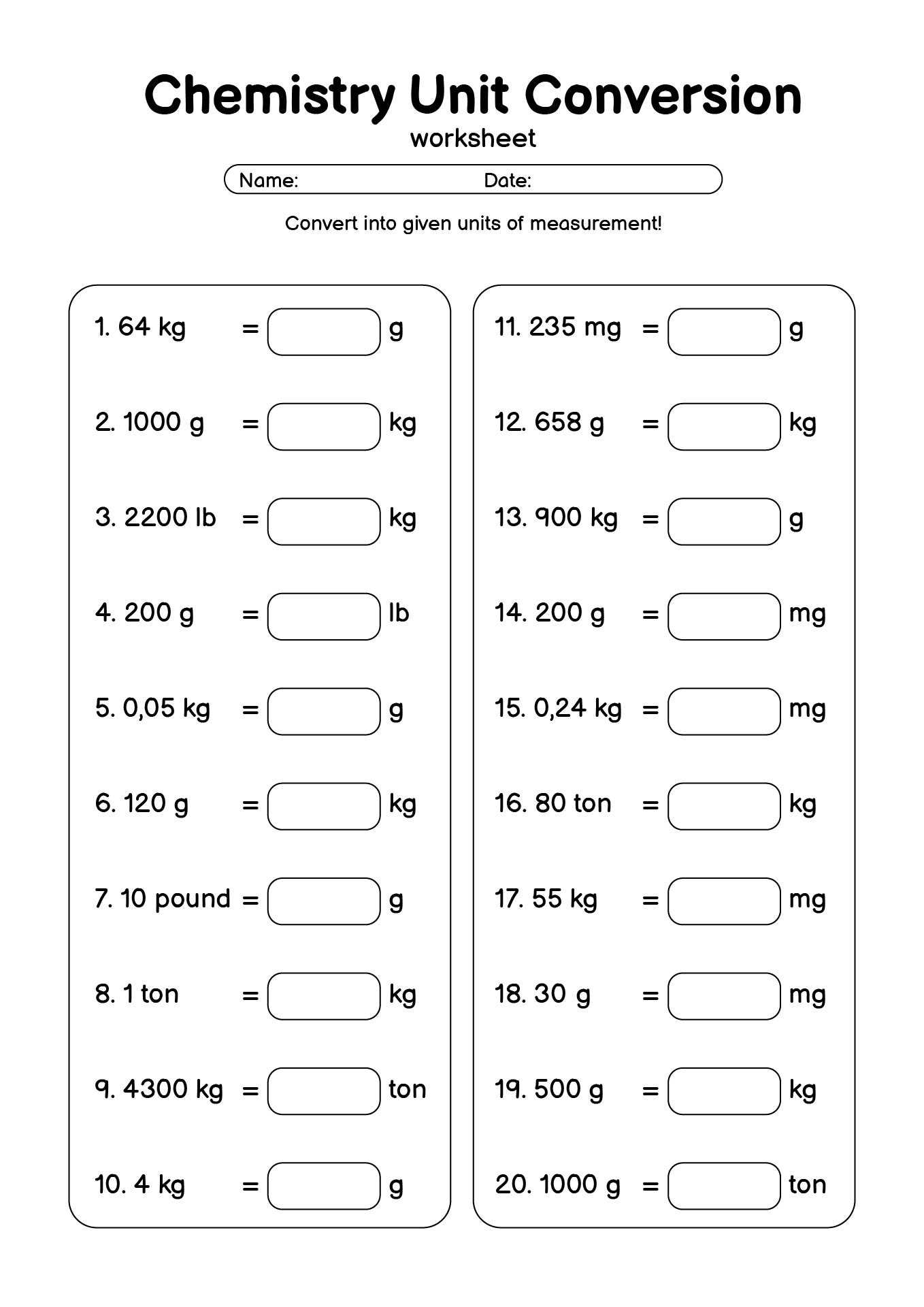
- Chemistry Unit Conversion Practice Worksheet
- Chemistry Conversion Factors Practice Worksheet
- Basic Chemistry Unit Conversion Worksheet
- Chemistry Unit Conversion Practice Worksheet
- Basic Chemistry Conversion Factors Sheet
- High School Chemistry Conversion Exercises
- Introductory Chemistry Conversion Worksheet
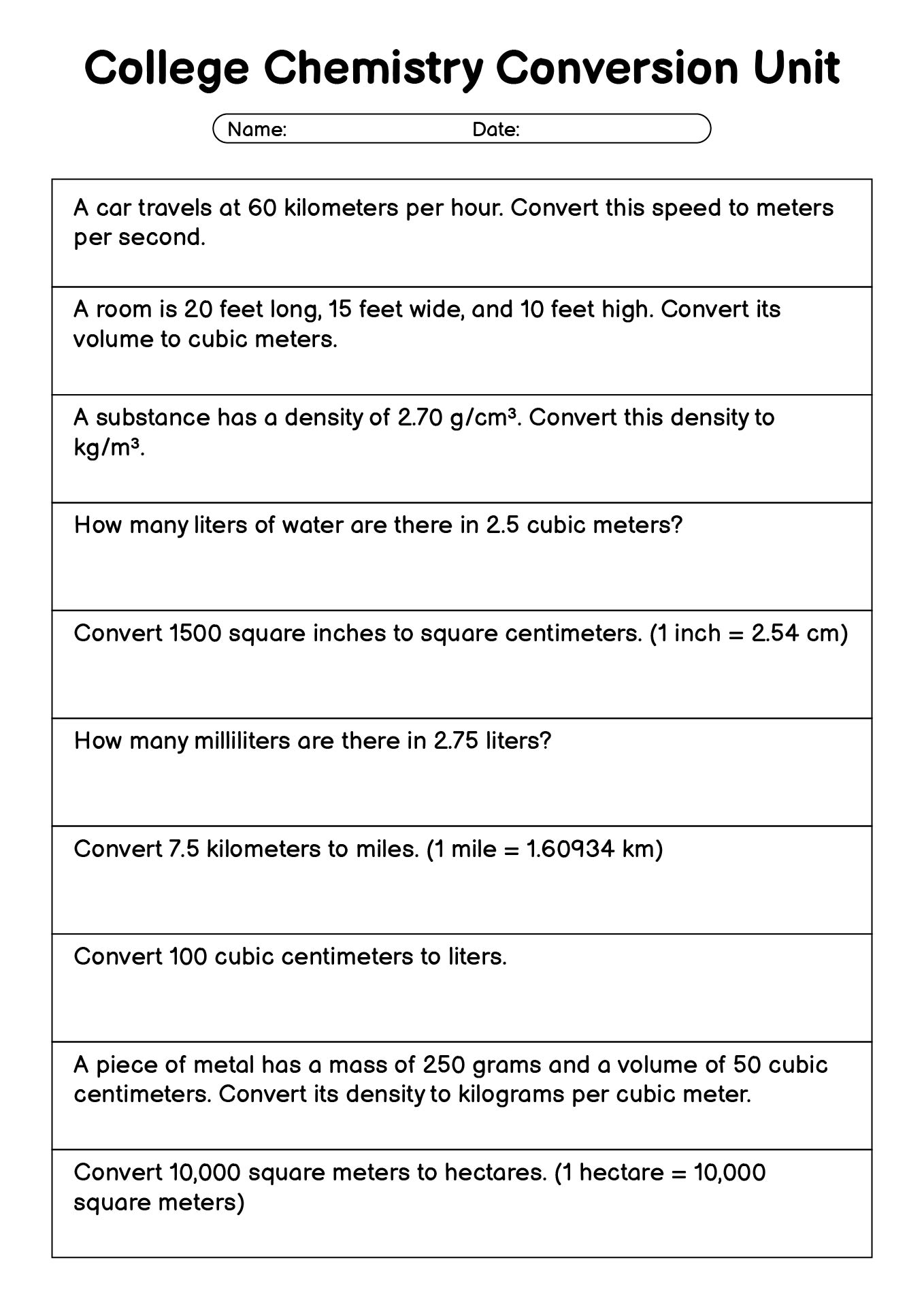
- College Chemistry Unit Conversion Problems
- General Chemistry Conversion Factors Challenge
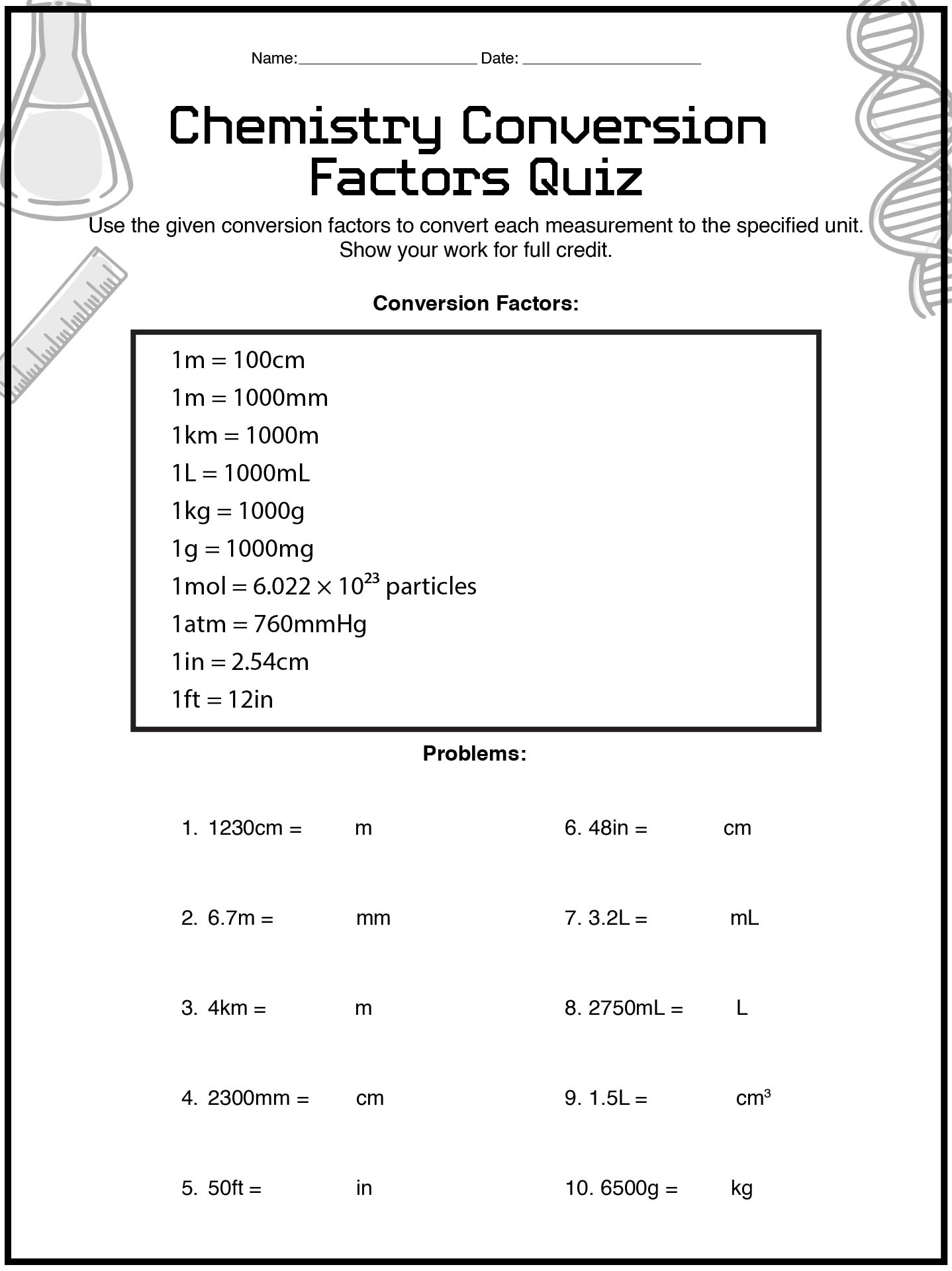
- Chemistry Conversion Factors Quiz Worksheet
- General Chemistry Conversion Factors Test Worksheet

More Worksheets
What Is a Chemistry Conversion Factors Worksheet?
A Chemistry Conversion Factors printable sheet is a structured practice tool that helps students convert measurements using ratios and units in a clear, organized format. Instead of memorizing formulas without context, students learn how to set up problems logically and follow a consistent process.
Typically, these worksheets guide learners through:
- Identifying the given value and unit
- Choosing the correct conversion factor
- Setting up dimensional analysis properly
- Canceling units step by step
- Checking whether the final unit makes sense
Many teachers prefer using a Chemistry Conversion Factors Worksheet PDF because it’s easy to print, reuse, and share across classes. It also pairs well with a chemistry conversion factors cheat sheet, allowing students to focus on the process rather than scrambling for reference information.
How Does a Chemistry Conversion Factors Worksheet Help Students Track Units Step by Step?
One of the biggest struggles in chemistry is keeping track of units. A Chemistry Conversion Factors printable sheet solves this by making units impossible to ignore. Students don’t just write numbers, they write units at every step.
This approach helps learners:
- Visually see which units cancel out
- Understand why certain conversion factors are used
- Catch mistakes before reaching a final answer
- Build habits that transfer to more advanced topics
Instead of guessing or skipping steps, students learn to slow down and think. This is especially helpful during chemistry conversion factors practice, where repetition builds confidence.
For teachers, this means fewer “I don’t know where I went wrong” questions. You can look at the setup and immediately spot the issue.
What Types of Problems Work Best with a Chemistry Conversion Factors Worksheet?
A Chemistry Conversion Factors printable sheet is flexible and works well with many common chemistry problems, especially those that require clear unit handling.
Some examples include:
- Metric unit conversions (grams to kilograms, milliliters to liters)
- Time and temperature-related conversions
- Density calculations
- Mole-to-mass conversions
- Multi-step word problems
Because the format encourages organized thinking, these conversion factors worksheets are ideal for both introductory lessons and review sessions. Many educators use them as guided practice before quizzes or as independent work after a lesson.
How Does a Chemistry Conversion Factors Worksheet Support Dimensional Analysis Skills?
Dimensional analysis can feel overwhelming at first, but a Chemistry Conversion Factors printable sheet breaks it into manageable steps. Instead of presenting dimensional analysis as a single intimidating method, the worksheet guides students through it gradually.
This helps students learn to:
- Set up equations correctly from the start
- Understand why units cancel mathematically
- See dimensional analysis as a reasoning tool, not a trick
- Apply the same logic across different problem types
Over time, students stop seeing conversions as random steps and start recognizing patterns. This makes advanced chemistry topics feel more approachable.
How Does a Chemistry Conversion Factors Worksheet Encourage Logical Problem Setup?
Chemistry Conversion Factors sheets encourage logical problem setup by teaching students to slow down and plan before calculating. Instead of jumping straight into math, the worksheet prompts learners to identify the given value, write the target unit, and select the correct conversion factors in the right order.
By organizing work step by step, students learn to line up units so unwanted units cancel cleanly. This visual structure makes mistakes easier to spot and correct.
How Does Our Chemistry Conversion Factors Worksheet Help Connect Math and Chemistry?
Many students struggle not because they dislike chemistry, but because they’re unsure how math fits into it. Our printable Chemistry Conversion Factors sheet is designed to bridge that gap.
Instead of separating calculations from concepts, the worksheet encourages students to:
- Read problems carefully
- Translate words into mathematical relationships
- Use ratios in a meaningful way
- Check answers using units, not just numbers
This approach builds confidence in both subjects at once. Students begin to trust the process instead of second-guessing every step.
So, Why Are Our Chemistry Conversion Factors Worksheets a Smart Addition to Any Chemistry Toolkit?
Our Chemistry Conversion Factors Printable Worksheets were created with real classrooms and busy households in mind. It’s not just about practice, it’s about making learning smoother for everyone involved.
Here’s why educators and parents find them useful:
- Clear layouts that reduce cognitive overload
- Consistent structure that students recognize quickly
- Easy pairing with notes or a chemistry conversion factors cheat sheet
- Suitable for classwork, homework, or review
- Simple to reuse year after year
So, if your students struggle with unit conversions, the right practice tool can make all the difference. Our Chemistry Conversion Factors Worksheet focuses on clarity, helping learners understand not just the answer, but the reasoning behind it. By breaking dimensional analysis into manageable steps, students learn how to track units, apply conversion factors, and solve problems with intention.
Download our printable Chemistry Conversion Factors Worksheet today and give your students or children a structured, supportive resource that helps chemistry feel logical and achievable!
Have something to share?
Popular Categories
Who is Worksheeto?
At Worksheeto, we are committed to delivering an extensive and varied portfolio of superior quality worksheets, designed to address the educational demands of students, educators, and parents.






















Comments