First Grade Science Worksheets Matter
Are you in search of engaging and educational resources to help your first-grade students grasp the concept of matter in science? Look no further! Our carefully crafted First Grade Science Worksheets on Matter are designed to captivate young minds, providing them with a solid foundation in understanding the properties and states of matter. With a variety of interactive activities and colorful illustrations, these worksheets will engage your students and make learning about matter fun and effortless. Get ready to inspire curiosity and spark a love for science in your classroom!
Table of Images 👆
- Science Properties of Matter Worksheets
- Properties of Matter Foldable
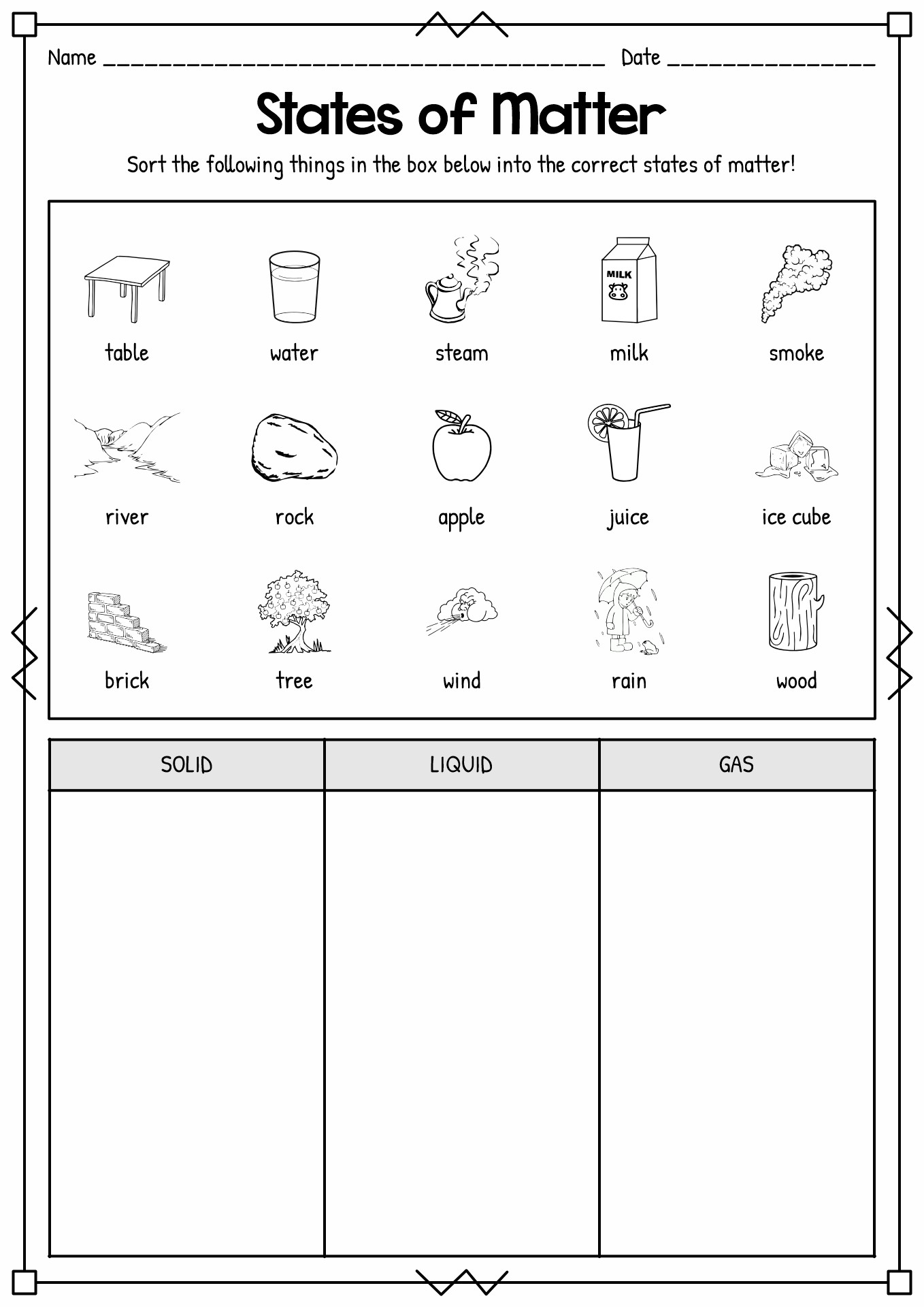
- Solids Liquids and Gases Worksheets
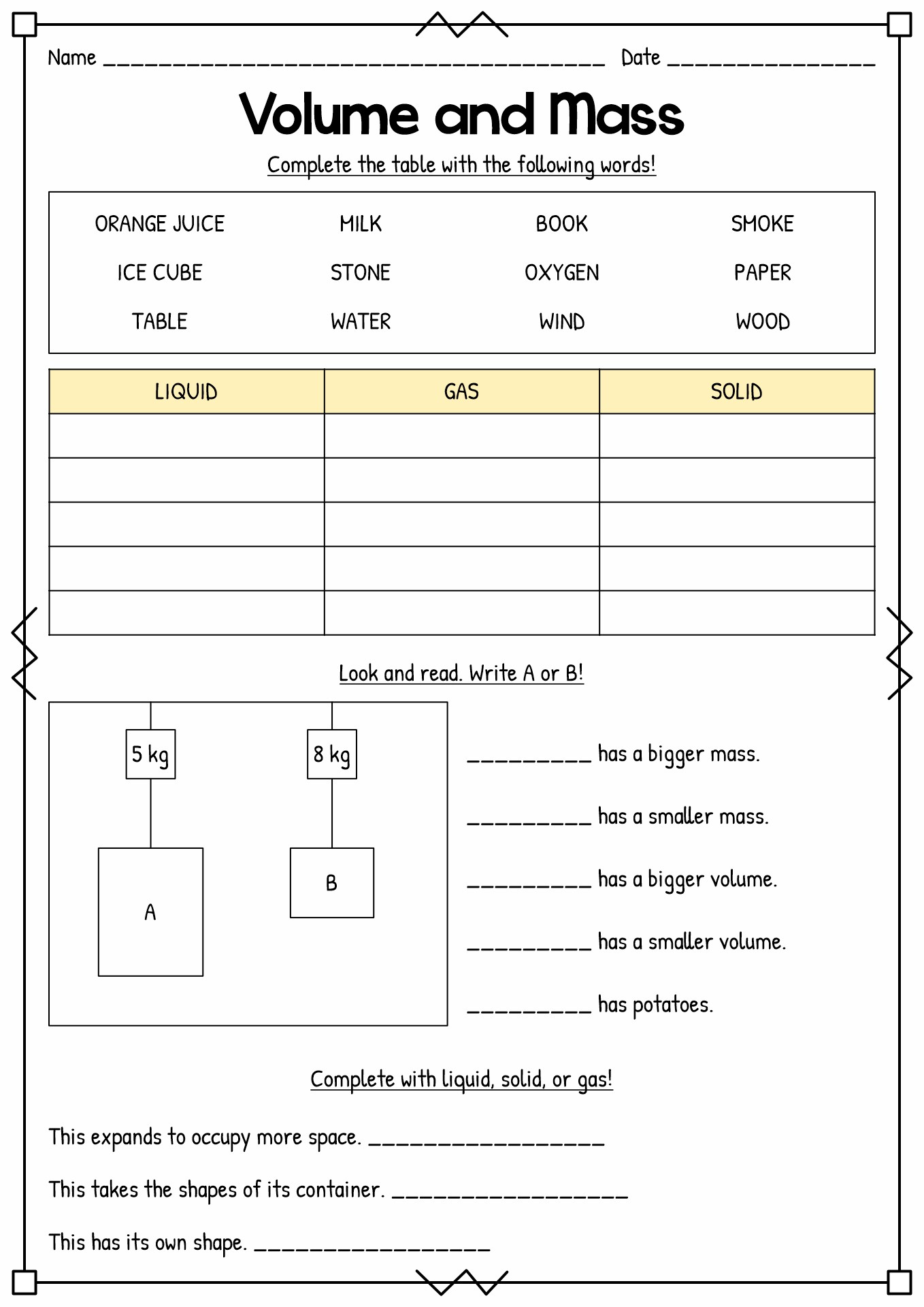
- 3rd Grade Science Volume Mass Liquid
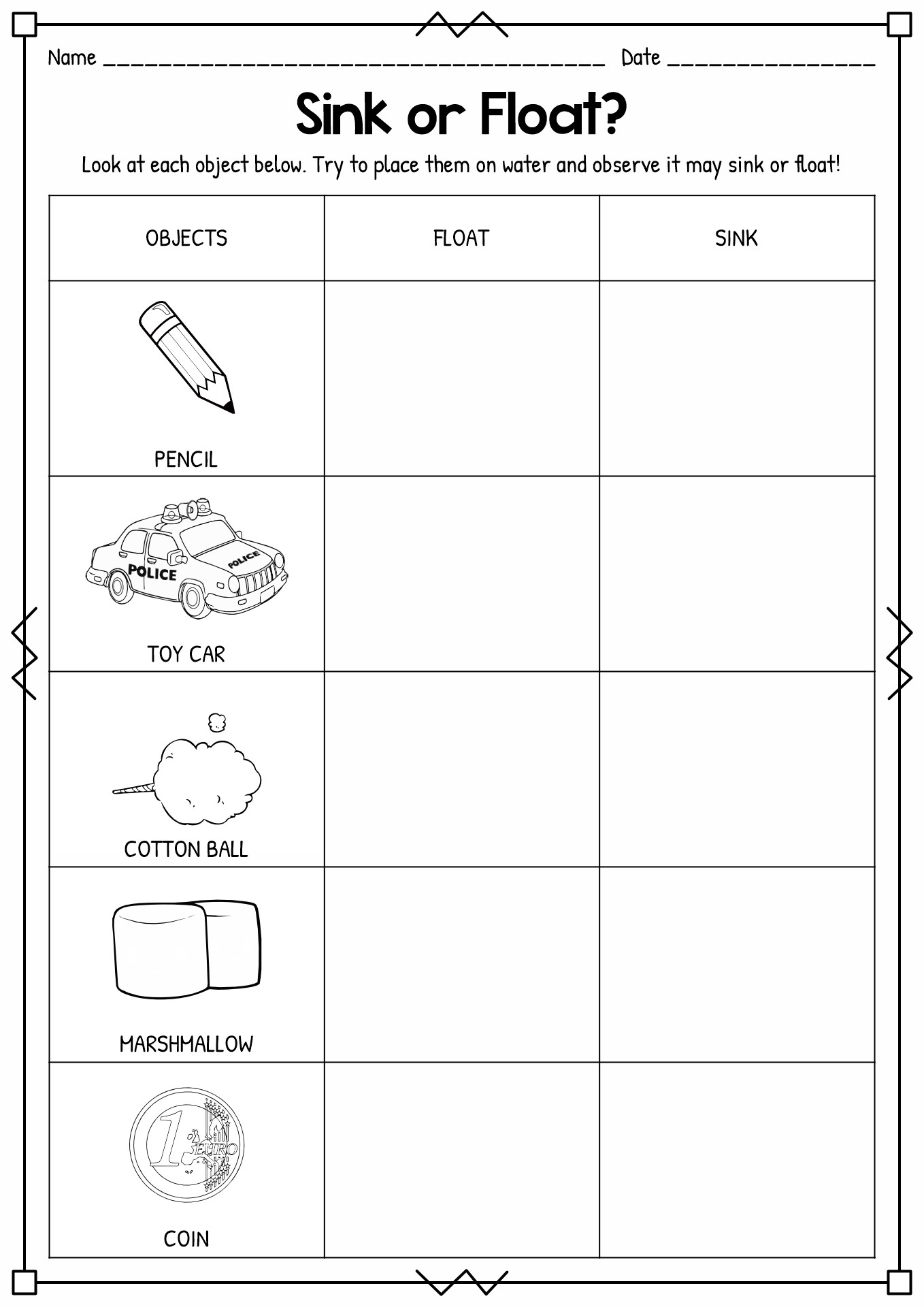
- Sink or Float Activity Worksheet
- First Grade Science Worksheets
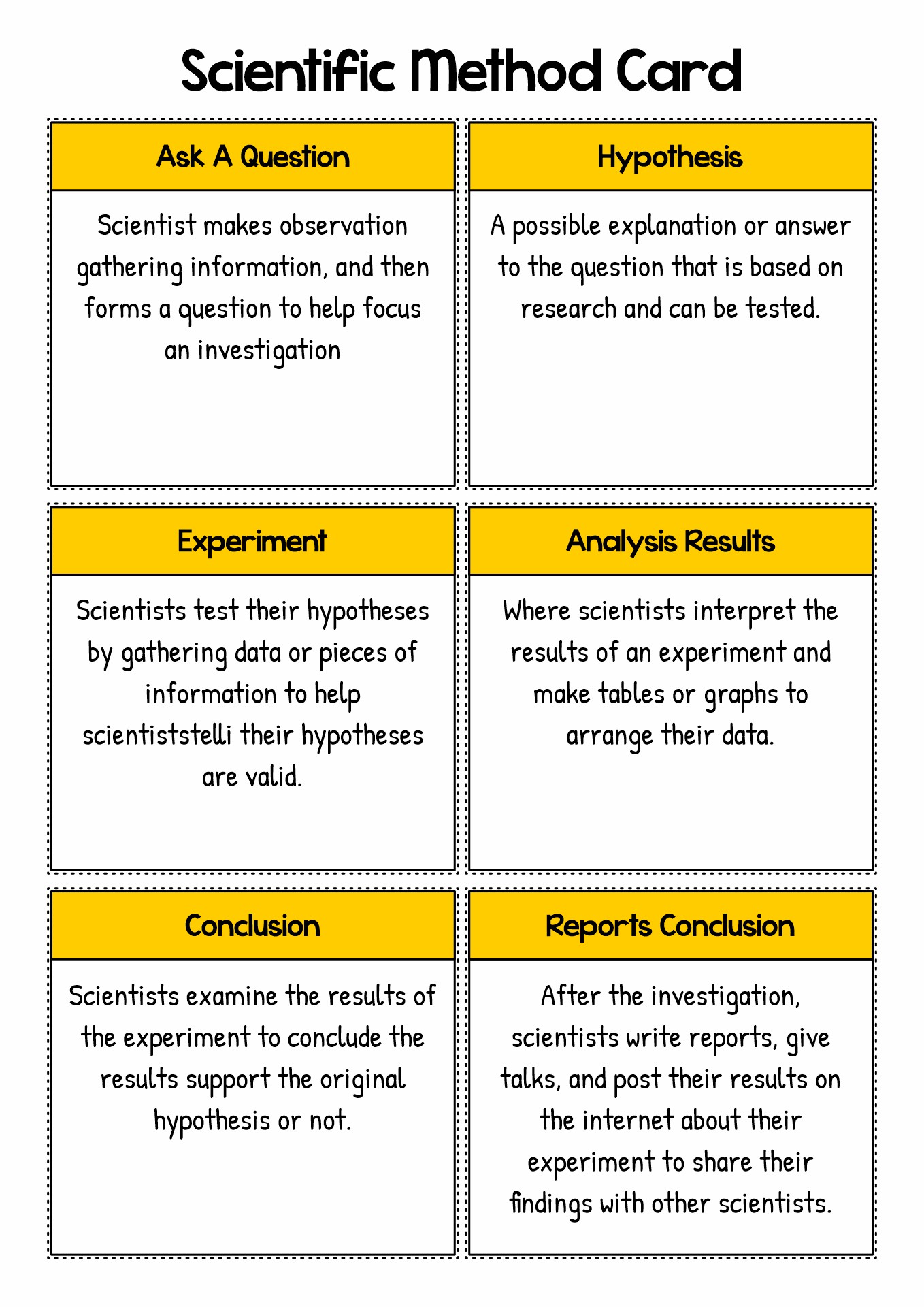
- Steps Scientific Method Cards
- Preschool Snowman Sequencing Worksheet
- 3rd Grade Math Assessment Test
- Plant Word Search Printable
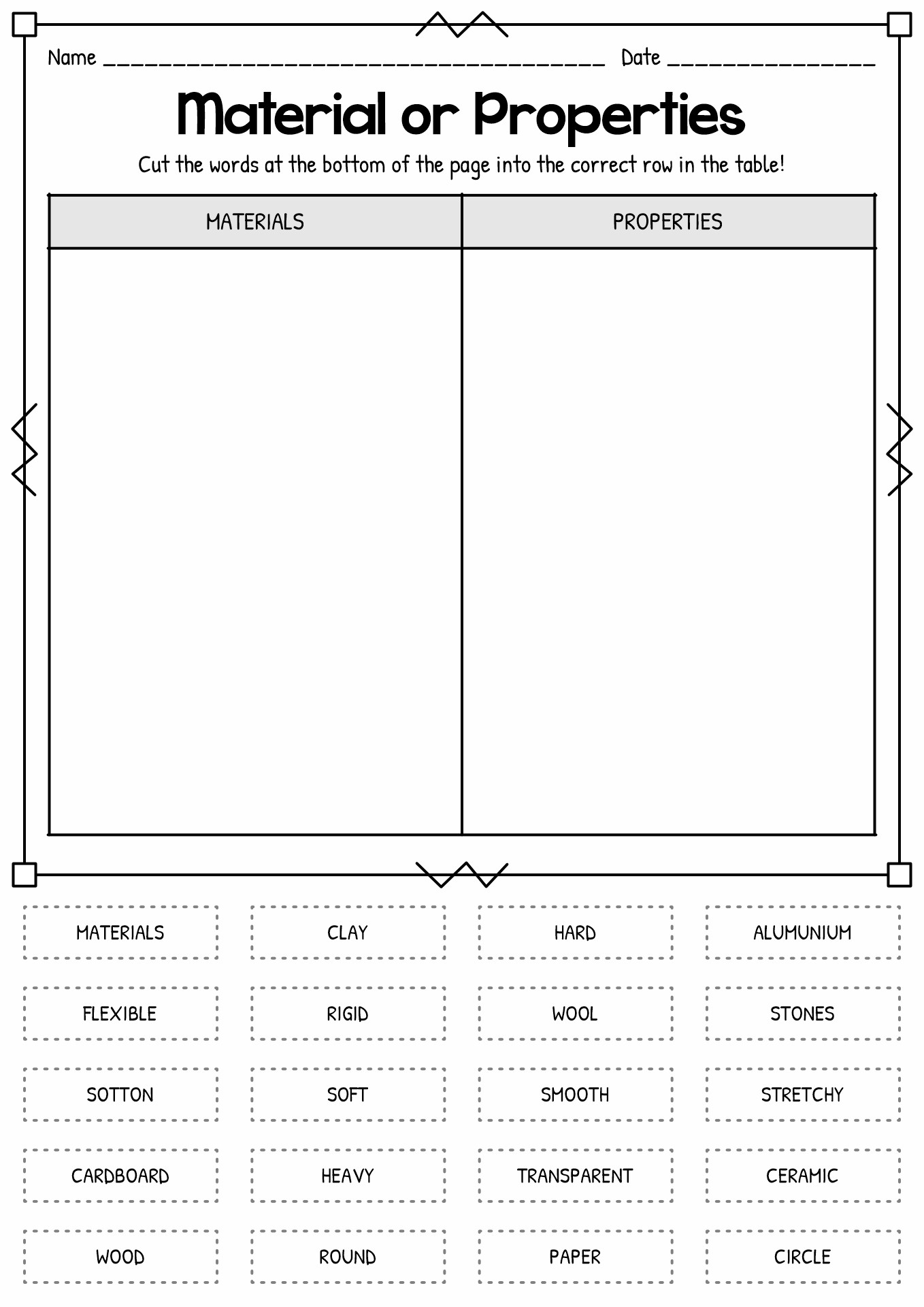
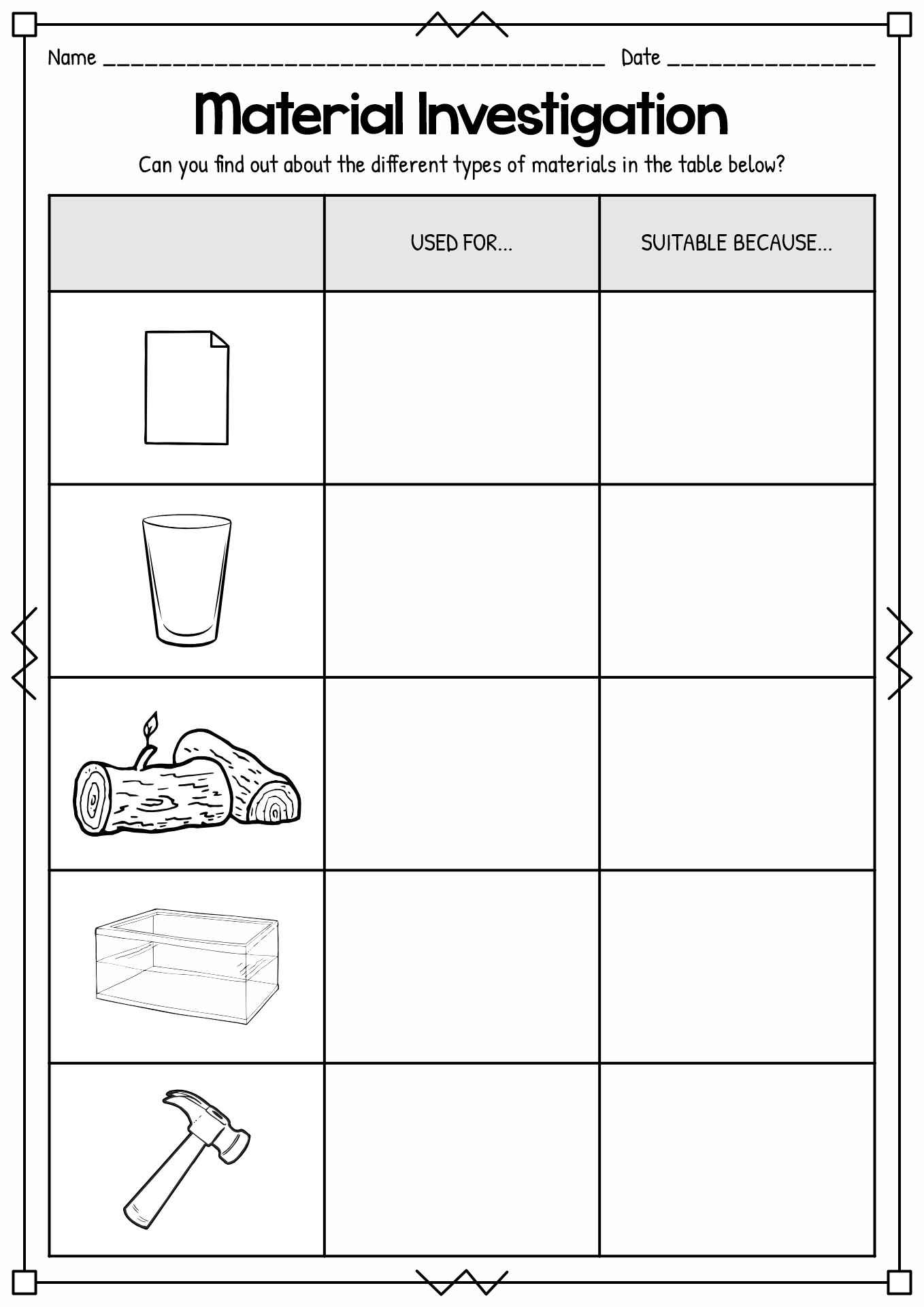
- Materials Science Activity Center
- Materials Science Activity Center

Understanding the states of matter can be fun for first graders, especially with our First Grade Science Worksheets Matter, which offer engaging and educational material.
More 1st Grade Worksheets
First Grade Reading Comprehension WorksheetsFirst Grade Reading Comprehension Worksheets
Telling Time Worksheets for First Grade
First Grade Clock Worksheets Printables
Writing Worksheets for 1st Graders
Easy 1st Grade Math Worksheets
Math Worksheets Subtraction 1st Grade
For First Grade Addition Worksheets
Plural Nouns Worksheets 1st Grade
Irregular Plurals Worksheets 1st Grade
Learn your surroundings through these First Grade Science Worksheets Matter!
Summary: Matter is the substance that assembles the universe and all the elements within it. Matter consists of many tiny particles known as atoms. It can undergo physical and chemical transformations. The tangible changes do not change the chemical arrangement of a substance, such as changes in state (melting, boiling) or change the shape and size. In science, there are three types states of matter, solid, liquid, and gas.
What is Matter in Science?
In general definition, we can say matter is a physical substance that makes an object up. The Earth and everything around us consists of matters. According to the University of Hawaii, the matter is anything that fills the space, and we can weigh them. Hence, it should have volume and mass. It is the substance that assembles the universe and all the elements within it. Matter consists of many tiny particles known as atoms. They are the building blocks of matter. They blend to form molecules. This mix works in tandem to create various types of substances.
Matter can undergo physical and chemical transformations. The tangible changes do not change the chemical arrangement of a substance, such as changes in state (melting, boiling) or change the shape and size. However, the chemical changes involve the rearrangement of atoms, creating new substances with different chemical properties. The study of matter is essential for various scientific disciplines' development, including physics and chemistry. Scientists have classified them into different types and have developed models and approaches to define their behaviour and properties.
How Many States of Matter Are There?
As we learned in the previous paragraph, matter can change. It can go from one phase to another. In science, there are three types states of matter, solid, liquid, and gas. Solid is a phase of substance when the particles inside it are packed and tied together with almost no movement. In this state, the atom's electrons only move a little and cause the substance to have low kinetic energy. It also causes the matter to hold a tight density. Solid substances have a concrete shape with definite mass and volume. In liquid states, the particles inside the material move more loosely and can float around each other. These movements cause the matter not to have a definite figure; instead, it will conform to the shape of the container. The liquid matter has a low density and is tough to compress. Lastly, the particles in the gas substance have a wide gap in between. It causes the element to have a high kinetic energy. In the gas states, matter does not have a definite shape, which makes them have a great deal of space. Based on National Geographic, there are other states in the universe; however, they rarely exist on Earth, such as plasma. It consists of particles with high charge and kinetic energy.
What are the Properties of Matter in Science?
According to the Online Consortium of Oklahoma, there are two classes of properties of matter, extensive and intensive. Intensive property means the characteristic that does not depend on the amount of the substance. Meanwhile, extensive property means the traits depend on the measured amount of the element. There are also physical and chemical properties of matter in science. Physical property means the aspects we can recognize without altering the chemical identity. Some examples of them are colour (intensive), density (intensity), boiling and melting point (intensive), volume (extensive), and mass (extensive). Meanwhile, chemical property means the traits we can determine by changing the molecular structure. The heat of combustion, chemical stability, flame ability and preferred oxidation are some examples of chemical properties.
Why is Learning Matter in Science Important?
Studying matters and everything around it is essential for students in the early year. It gives them an introduction to their surrounding and helps them understand the events occurring around them. Many experts agree that learning about states of matter will be a solid foundation for young students to learn about chemistry and physics. It is also the door for the students to understand the characteristics of the substance around them. This knowledge will help the young learners to survive daily life while utilizing the matter around them. Studying matter is also crucial to develop research and findings to help humans increase their quality of life by the scientist.
Who Discovered States of Matter?
The study of matter in science is crucial for humanity, as it helps us in many daily life cases. The discovery of this knowledge starts in the Ancient Greek era, around 600 or 500 BCE. They believe that the altering phase in the water element is the single principle of the universe. Even though this statement is untrue based on today's science, it is the start of the current theory of matter. The scientist agrees that plasma is the first matter in the universe. It is the beginning of how the universe and everything within happened.
Have something to share?
Popular Categories
Who is Worksheeto?
At Worksheeto, we are committed to delivering an extensive and varied portfolio of superior quality worksheets, designed to address the educational demands of students, educators, and parents.
























Comments
Printable images on first grade science worksheets matter because they visually enhance comprehension and make learning more engaging for young students, reinforcing the concepts taught in the curriculum.
The First Grade Science Worksheets Matter are a helpful tool for sparking curiosity and reinforcing concepts in an engaging way!
I love using these First Grade Science Worksheets Matter with my young learners! They are a great tool to introduce essential concepts in a simple and engaging way. Thank you for providing such a valuable resource!
I found the First Grade Science Worksheets Matter to be a helpful tool for my child's learning journey. The worksheets provided a structured and engaging way to explore and understand the concept of matter. Thank you for this printable resource!