Word Equations Worksheet Answer
Are you struggling to find the answers to your word equations worksheet? Look no further! In today's blog post, we will provide you with a comprehensive answer key to help you solve any word equations that you may be having trouble with. Whether you're a student studying chemistry or a teacher looking for additional resources to support your lesson plans, this worksheet answer will help you understand the concept of word equations and provide a valuable tool for learning and practice.
Table of Images 👆
- Algebra Solving Linear Equations Worksheets
- Balancing Chemical Equations Worksheet Answers
- 7th Grade Math Worksheets Algebra
- 7th Grade Math Worksheets
- Graphing Linear Equations Using Intercepts
- Graphing Quadratic Functions Worksheet Answers
- Bonding Worksheet Answer Key
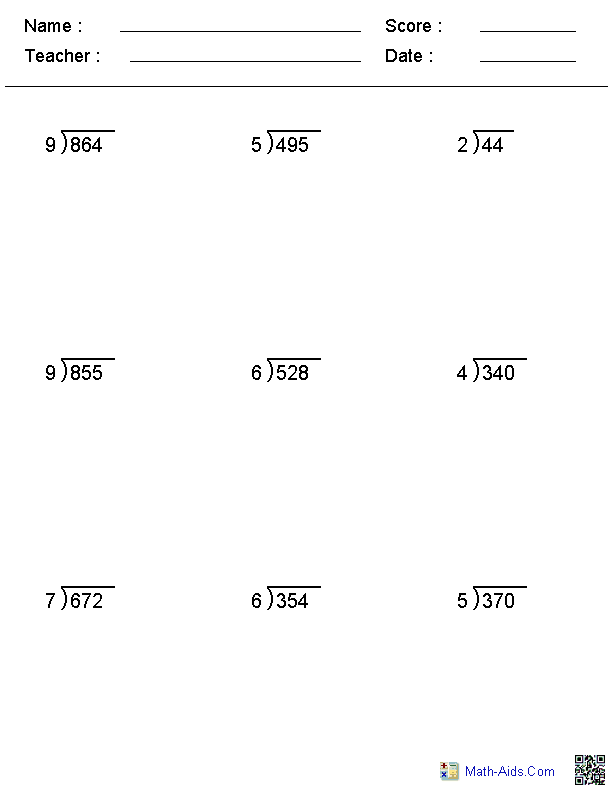
- Grade Long Division Worksheet
- Algebra 1 Worksheets
- 3 Factor Multiplication Worksheets
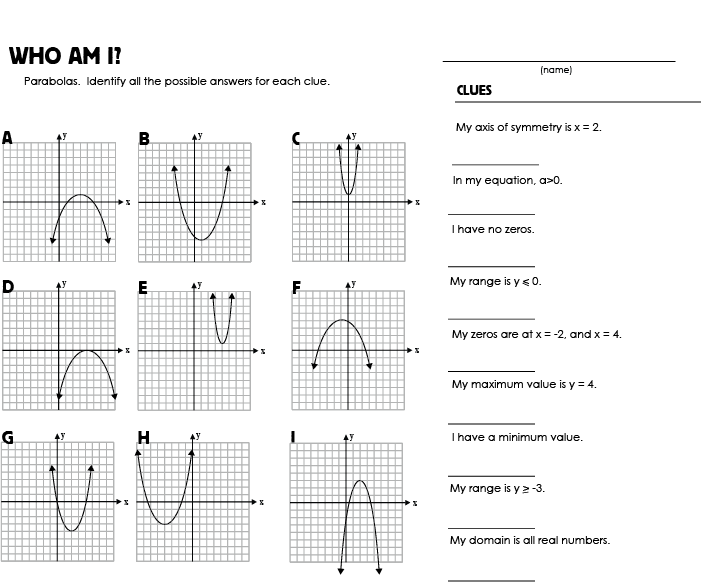
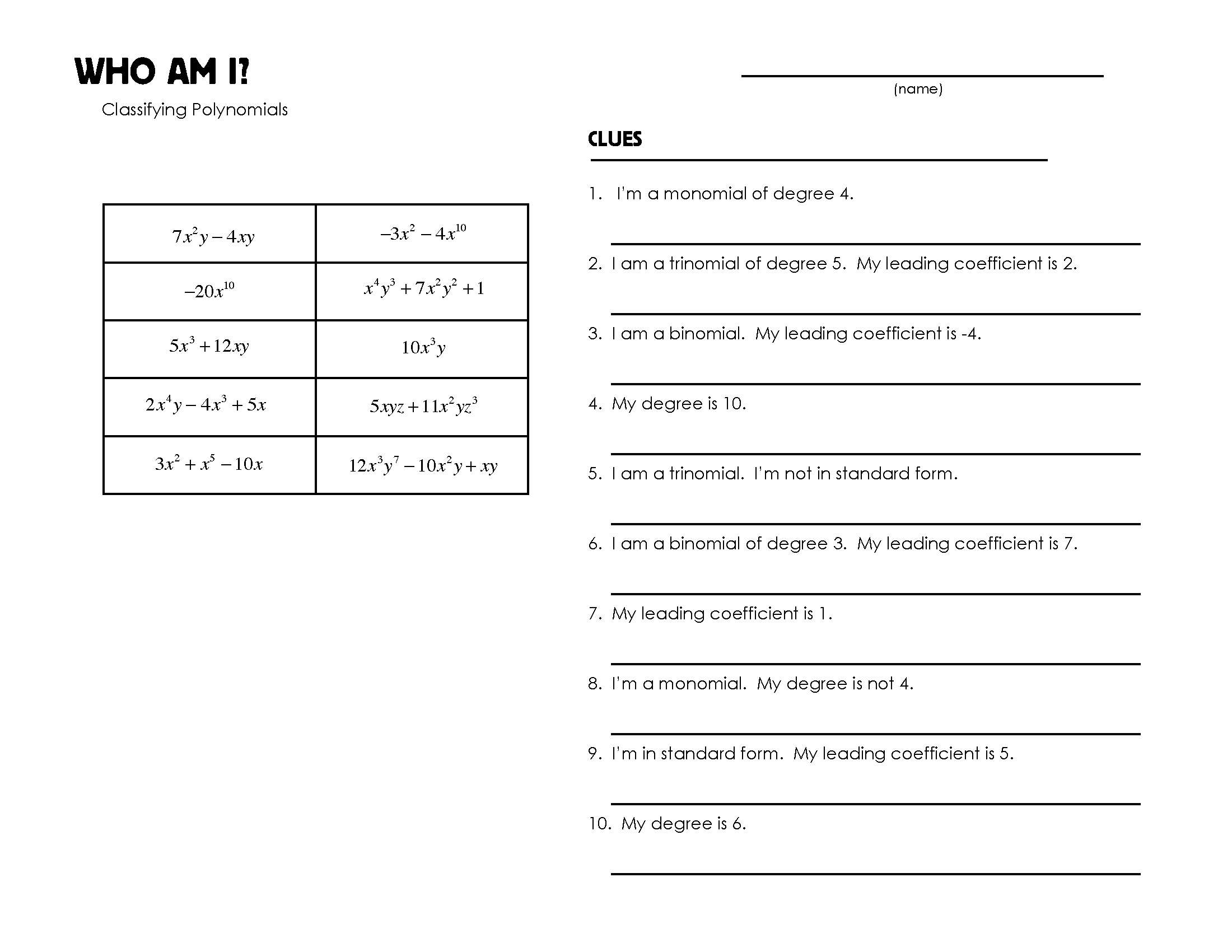
- Who AM I Worksheet Answers
- Decimal Word Problems
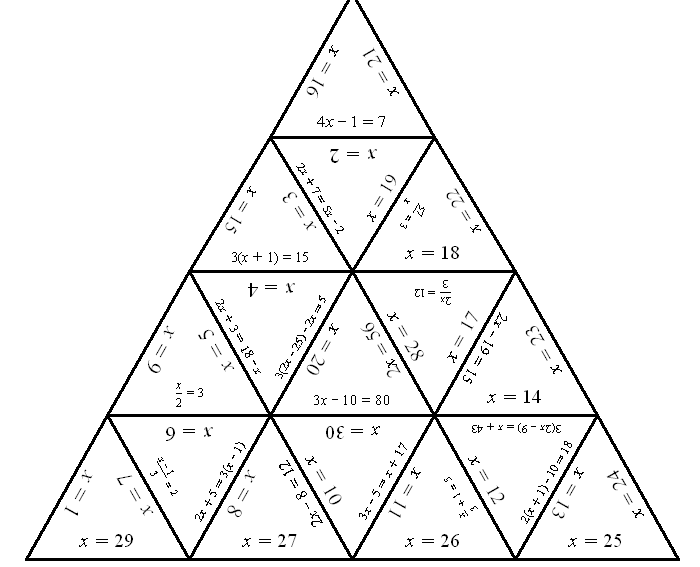
- Tarsia Puzzles
- Periodic Table with Element Mass
More Word Worksheets
Practice Writing Words WorksheetsSpelling Words Worksheets Grade 2
Have Sight Word Worksheet
Fry's First 100 Words Worksheets
First 100 Sight Words Printable Worksheets
Blending Words Worksheets for Kindergarten
9th Grade Worksheets Spelling Words
Matching Definitions to Words Worksheets
Sight Words Worksheets 5th Grade
Element Word Search Worksheet
What is a word equation?
A word equation is a written representation of a chemical reaction using words instead of chemical formulas. It describes the reactants and products involved in the reaction and provides a simple way to understand the overall process without detailed chemical notation.
How are word equations different from chemical equations?
Word equations are written in words or a descriptive form, while chemical equations are written using chemical symbols and formulas to represent the reactants and products of a chemical reaction. Chemical equations provide specific information about the number and type of atoms or molecules involved in a reaction, while word equations provide a more general description of the reaction. Chemical equations are more precise and detailed, making them essential in chemistry for accurately representing chemical reactions.
What is the purpose of balancing a word equation?
The purpose of balancing a word equation is to ensure that the equation obeys the law of conservation of mass. Balancing a word equation involves adjusting the number of atoms of each element on both sides of the equation so that the same number of each type of atom is present on both sides, reflecting that matter is neither created nor destroyed in a chemical reaction.
How are reactants and products represented in a word equation?
Reactants are typically listed on the left side of a word equation, separated by a plus sign (+), while products are listed on the right side, also separated by a plus sign. For example, in the word equation "hydrogen + oxygen ? water," hydrogen and oxygen are the reactants on the left side, and water is the product on the right side.
What does a balanced word equation indicate?
A balanced word equation indicates that the number of atoms of each element is the same on both sides of the equation, ensuring that mass is conserved during a chemical reaction. It shows the reactants and products involved in the reaction and the stoichiometry of the reaction, providing a clear representation of the chemical change taking place.
How can you determine the number of atoms in a chemical formula from a word equation?
To determine the number of atoms in a chemical formula from a word equation, you need to first balance the equation and then write down the corresponding chemical formula for each substance involved. Next, you can use the subscripts in the chemical formulas to determine the number of atoms for each element present in the formula. Count the number of atoms for each element by multiplying the subscript with the coefficient in front of the chemical formula in the balanced equation. This will give you the total number of atoms for each element in the chemical formula.
What is the role of coefficients in balancing word equations?
The coefficients in balancing word equations represent the relative amounts of each element or compound involved in a chemical reaction. By adjusting the coefficients in front of the chemical formulas, we ensure that the law of conservation of mass is satisfied, meaning that the total number of atoms of each element on both sides of the equation is equal. This balancing process helps us understand the stoichiometry of the reaction and determine the correct ratio in which reactants combine and products are formed.
What is the significance of a double arrow in a word equation?
A double arrow in a word equation indicates a reversible reaction, meaning that the reactants can form products but the products can also react to form the original reactants. It symbolizes that the reaction can proceed in both the forward and reverse directions.
How do word equations help predict the outcome of a chemical reaction?
Word equations help predict the outcome of a chemical reaction by providing a simple and concise representation of the reactants and products involved in the reaction. By writing out a word equation, one can easily identify the substances that will react with each other and the products that will be formed. This allows for a quick analysis of the reaction and helps in predicting the possible outcomes based on the known chemical properties of the substances involved.
Can a word equation alone provide all the information about a chemical reaction?
No, a word equation alone cannot provide all the information about a chemical reaction. While a word equation can describe the reactants and products involved in a reaction, it does not provide specific details such as the quantities of each substance involved, the conditions under which the reaction occurs, or the energy changes that take place. To fully understand a chemical reaction, additional information such as a balanced chemical equation, reaction conditions, and reaction mechanisms may be needed.
Have something to share?
Who is Worksheeto?
At Worksheeto, we are committed to delivering an extensive and varied portfolio of superior quality worksheets, designed to address the educational demands of students, educators, and parents.



























Comments