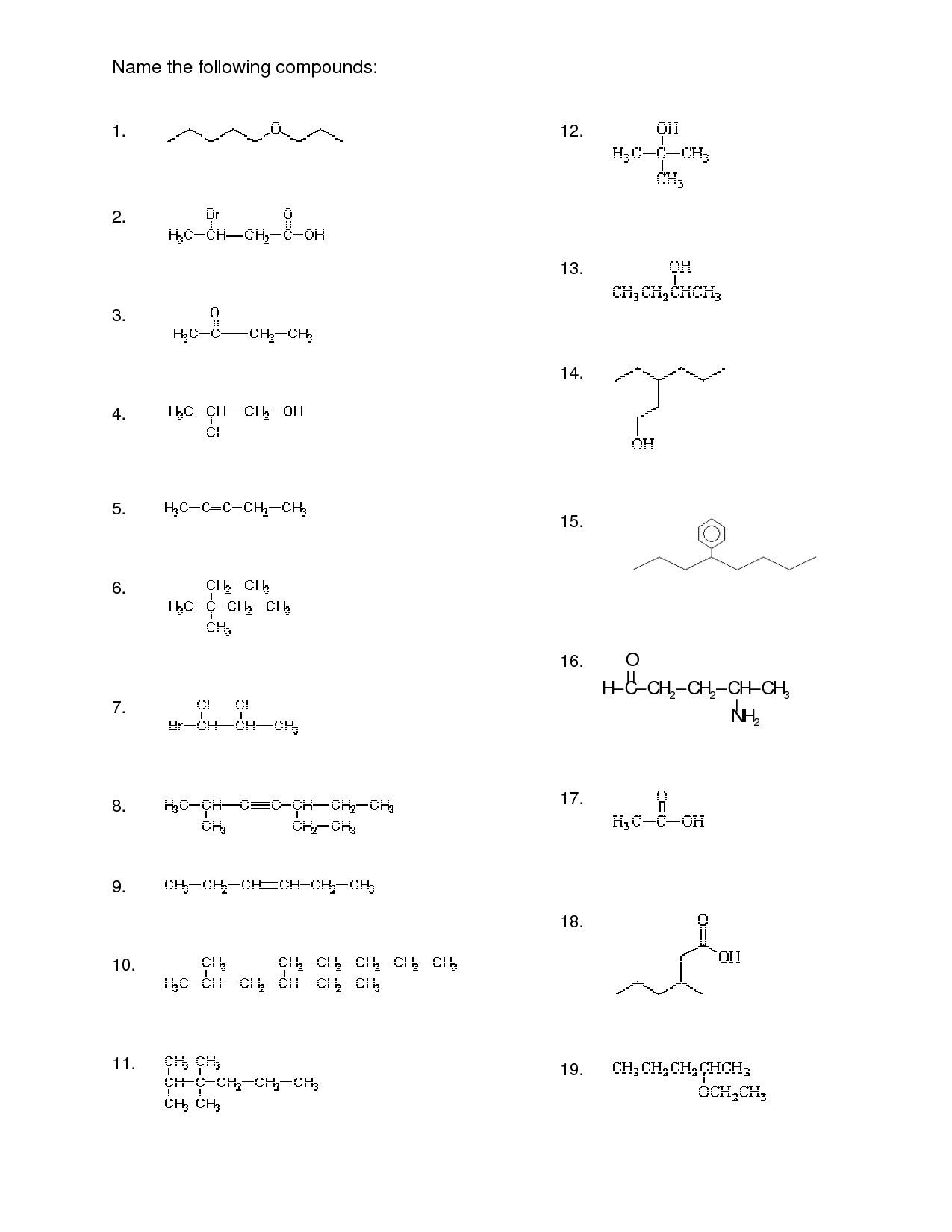
Organic Compound Nomenclature Worksheets
Organic compound nomenclature worksheets are a useful tool for those studying chemistry and looking to enhance their understanding of the naming and classification of organic compounds. These worksheets provide a comprehensive collection of exercises and practice problems aimed at honing one's skills in identifying the entity and subject of organic compounds. By engaging with these worksheets, learners can strengthen their proficiency in deciphering the complex naming conventions used in organic chemistry.
Table of Images 👆
- Organic Compounds Worksheet
- Naming Organic Compounds Worksheet
- Molecular Compounds Worksheet
- Organic and Inorganic Compounds Worksheet
- Naming Organic Compounds Practice Worksheet
- Organic Functional Groups Worksheet
- Organic vs Inorganic Compounds Worksheet Answers
- Chemistry Naming Compounds Worksheet Answers
- Organic vs Inorganic Compounds Worksheet
- Organic Chemistry Naming Alkanes Worksheet
- Organic Chemistry Nomenclature Worksheets with Answers
- Naming Ionic Compounds Worksheet Answer Key
- Naming Covalent Compounds Worksheet
- Naming Organic Compounds Worksheet Answer
- Naming Compounds and Writing Formulas Worksheet
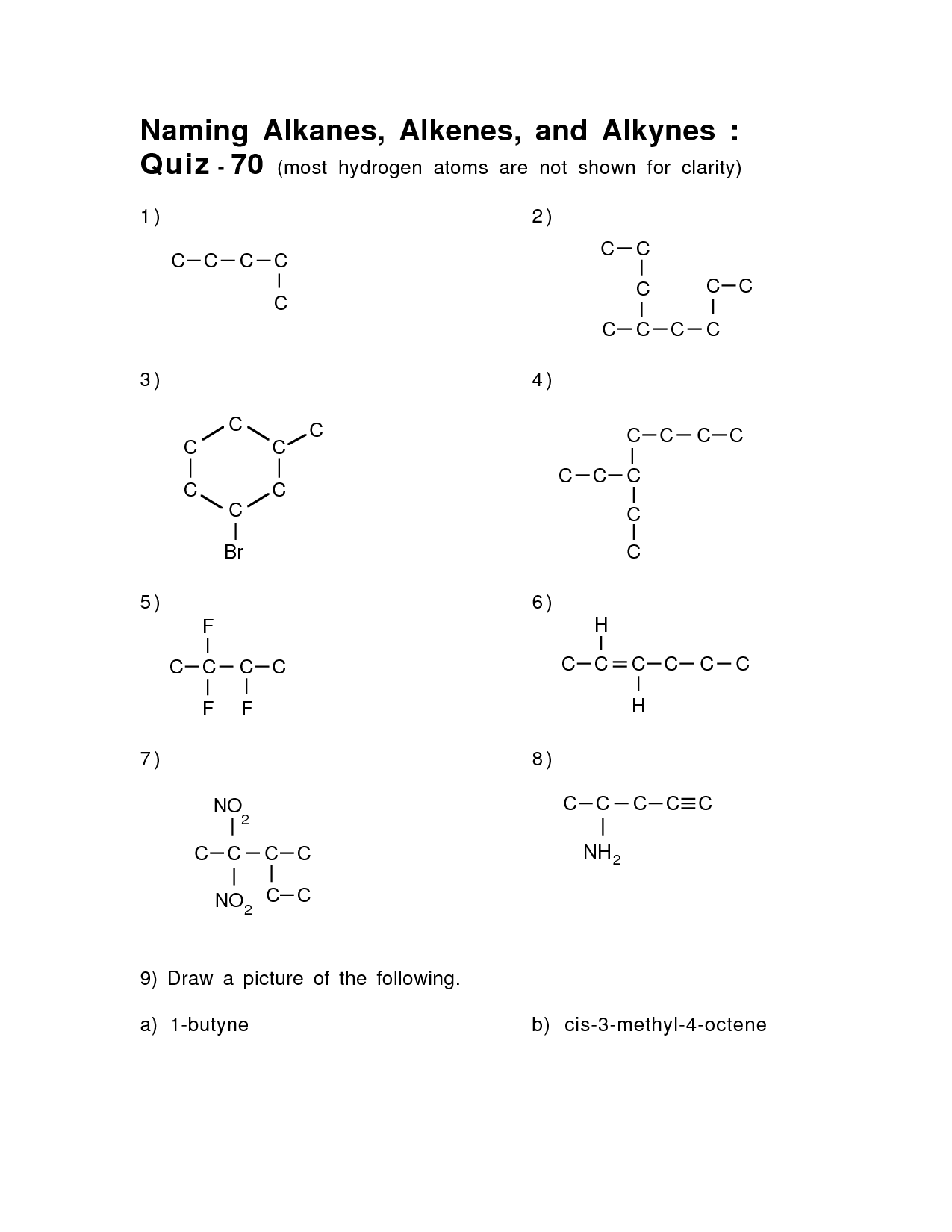
- Naming Alkanes Alkenes and Alkynes
- Organic Chemistry Functional Groups Worksheet
More Other Worksheets
Kindergarten Worksheet My RoomSpanish Verb Worksheets
Cooking Vocabulary Worksheet
DNA Code Worksheet
Meiosis Worksheet Answer Key
Art Handouts and Worksheets
7 Elements of Art Worksheets
All Amendment Worksheet
Symmetry Art Worksheets
Daily Meal Planning Worksheet
What is organic compound nomenclature?
Organic compound nomenclature refers to the systematic naming system used to identify and describe organic molecules based on their structure and composition. It involves naming the different parts of the molecule according to specific rules and conventions established by the International Union of Pure and Applied Chemistry (IUPAC) to ensure clarity, consistency, and precision in communication. By following these rules, chemists can accurately identify and differentiate between various organic compounds in a standardized and universally accepted manner.
What are the general rules for naming organic compounds?
The general rules for naming organic compounds involve identifying the longest continuous carbon chain (parent chain), numbering the carbons in the chain to give the lowest set of locants (numbers), naming and locating substituents on the parent chain, using prefixes to denote the number and type of substituents, and alphabetizing when multiple substituents are present. Additionally, the naming of organic compounds may also involve considering functional groups and suffixes to indicate the type of compound.
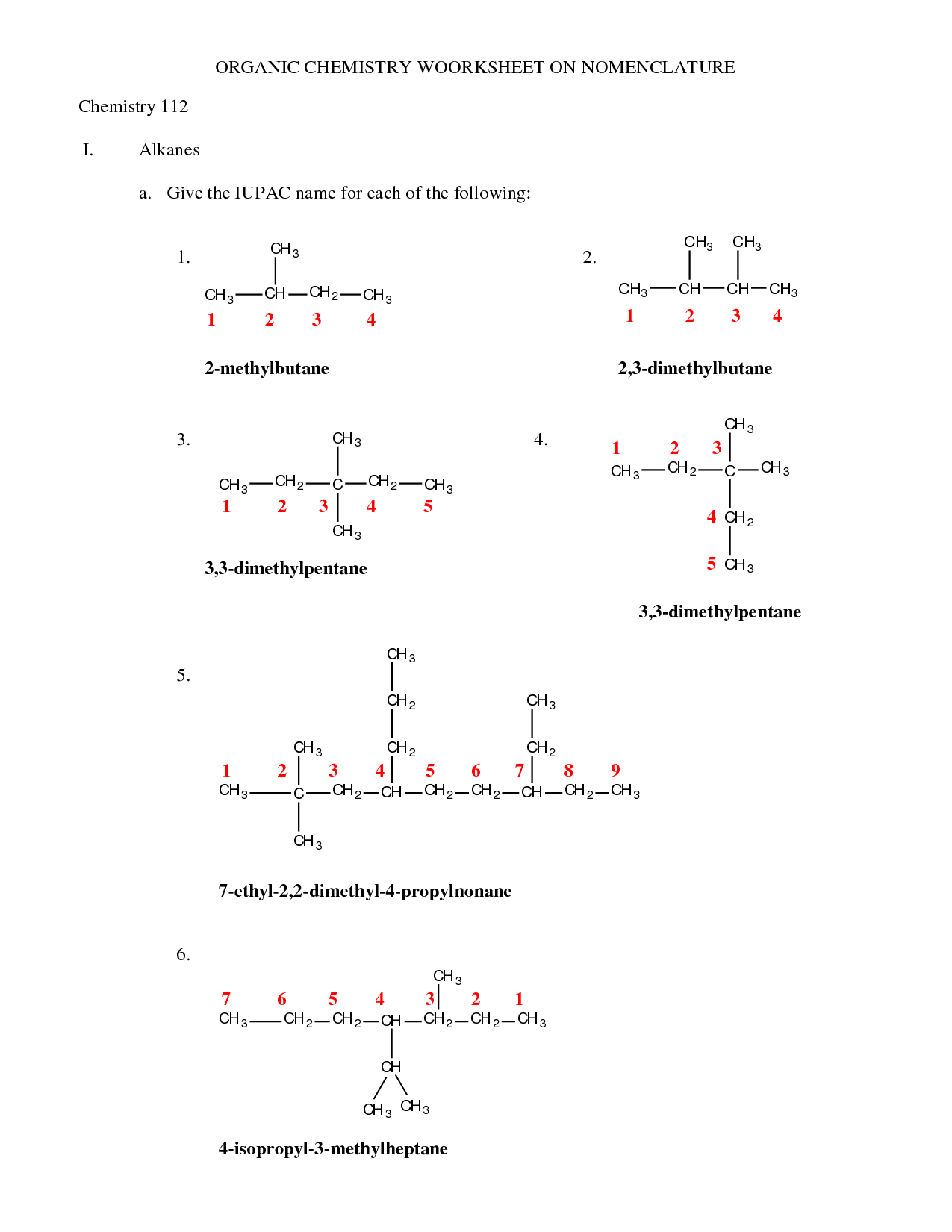
How do you determine the parent chain in a compound?
To determine the parent chain in a compound, look for the longest continuous carbon chain that contains the functional group with the highest priority. Typically, you would prioritize the chain that provides the most substituents on the parent chain, as long as it meets the structural requirements of the functional group. The parent chain is named based on the number of carbon atoms it contains, with the suffix indicating the type of carbon-carbon bond present in the chain.
How do you identify and name functional groups in a compound?
To identify and name functional groups in a compound, you first need to identify the atoms present in the compound and then look for common functional group patterns. Common functional groups include hydroxyl (-OH), carbonyl (C=O), carboxyl (-COOH), amino (-NH2), and many others. Once you identify a functional group, you can name it based on its structure and position in the compound following IUPAC naming conventions. The functional group name is usually included as a prefix or suffix in the compound name to indicate its presence.
How do you number the carbon atoms in a compound?
Carbon atoms in a compound are typically numbered based on the longest carbon chain present in the molecule. The carbon closest to a functional group or branch is usually assigned the lowest number to give the substituents the smallest possible numbers. The numbering of carbon atoms is done systematically according to the nomenclature rules specified by the International Union of Pure and Applied Chemistry (IUPAC) to ensure consistency and clarity in naming organic compounds.
What are substituents and how do you name them?
Substituents are groups of atoms attached to a parent molecule that can alter its properties. To name substituents, you use prefixes based on the number of carbons in the chain of the substituent. For example, a methyl group (CH3) has one carbon, an ethyl group (C2H5) has two carbons, and so on. These prefixes are then added to the parent molecule's name as a prefix before the molecule name, indicating where the substituent is located on the parent molecule.
How do you indicate multiple substituents in a compound?
To indicate multiple substituents in a compound, you can use prefixes such as di- (for two substituents), tri- (for three substituents), tetra- (for four substituents), and so on before the substituent names. Additionally, you may need to use numerical locants to specify the position of each substituent on the parent compound's carbon chain. This naming convention helps to accurately describe and differentiate between the various substituents present in a compound.
How do you name compounds with multiple functional groups?
When naming compounds with multiple functional groups, you need to identify the principal functional group based on the order of precedence outlined in IUPAC nomenclature rules. The principal functional group is given the highest priority in naming the compound, while the other functional groups are named as substituents. Each substituent is prefixed with a locator number to indicate its position on the parent chain, and the substituents are listed in alphabetical order. Special prefixes like di-, tri-, tetra- are used if there is more than one of the same substituent. Overall, follow systematic naming rules and prioritize the principal functional group to correctly name compounds with multiple functional groups.
How do you handle cyclic compounds in nomenclature?
When naming cyclic compounds, the prefix "cyclo" is added before the parent hydrocarbon name to indicate that the molecule is a closed ring structure. The numbering of carbons in the ring starts at a substituent that is alphabetically first and is designed to give the substituents the lowest possible numbers. Any cycloalkanes with one substituent do not require a number to indicate the position of the substituent, but for those with multiple substituents, numbers must be used to locate the substituents on the carbon chain.
What are some common mistakes to avoid when naming organic compounds?
Some common mistakes to avoid when naming organic compounds include failing to prioritize functional groups in the name, neglecting to use the appropriate prefixes and suffixes to indicate the number and position of substituents, not following the IUPAC naming rules accurately, and overlooking the stereochemistry of chiral compounds. It's crucial to pay attention to detail and adhere to the systematic nomenclature guidelines to ensure clarity and accuracy in naming organic compounds.
Have something to share?
Who is Worksheeto?
At Worksheeto, we are committed to delivering an extensive and varied portfolio of superior quality worksheets, designed to address the educational demands of students, educators, and parents.

































Comments