Fluorine Worksheets Activity
Fluorine worksheets provide an engaging and interactive way for students to explore the properties and characteristics of this fascinating chemical element. Designed for middle school and high school students studying chemistry, these worksheets offer a wide range of activities that focus on the entity of fluorine and its role in various subjects within the field of chemistry.
Table of Images 👆
- Chemistry Periodic Table Puns Worksheet Answers
- Year 3 Maths Homework Sheet
- Personal Hygiene Lesson Plans
- Scientific Notation Worksheet Answers Chemistry
- Lewis and Clark Worksheets
- As Periodic Table of Elements
- Nuclear Decay Worksheet Answer Key
- Blank Tooth Outline Printable
- Periodic Table Worksheets
- Personal Dental Hygiene
- Dental Hygiene Lesson Plans
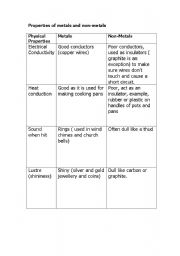
- Properties of Metals and Nonmetals Worksheet
- Sample Lesson Plan Examples
- Yoga Coloring Pages Printable
More Other Worksheets
Kindergarten Worksheet My RoomSpanish Verb Worksheets
Cooking Vocabulary Worksheet
DNA Code Worksheet
Meiosis Worksheet Answer Key
Art Handouts and Worksheets
7 Elements of Art Worksheets
All Amendment Worksheet
Symmetry Art Worksheets
Daily Meal Planning Worksheet
What is the atomic number of fluorine?
The atomic number of fluorine is 9.
What is the chemical symbol for fluorine?
The chemical symbol for fluorine is F.
What is the state of fluorine at room temperature?
Fluorine is a gas at room temperature, typically existing as a diatomic molecule with the chemical formula F2.
What is the boiling point of fluorine?
The boiling point of fluorine is -188.1 degrees Celsius.
What is the melting point of fluorine?
The melting point of fluorine is -219.67 degrees Celsius (-363.41 degrees Fahrenheit).
Is fluorine a metal, nonmetal, or metalloid?
Fluorine is a nonmetal.
What is the electron configuration of fluorine?
The electron configuration of fluorine is 1s2 2s2 2p5.
How many valence electrons does fluorine have?
Fluorine has 7 valence electrons.
Is fluorine a highly reactive element?
Yes, fluorine is a highly reactive element due to its strong electronegativity, meaning it readily forms compounds with other elements to achieve a stable configuration. It is the most electronegative and reactive of all the elements.
What are some common uses of fluorine in various industries?
Fluorine is commonly used in various industries such as pharmaceuticals for manufacturing fluorinated compounds, in the production of refrigerants and air conditioning systems, in the production of Teflon for non-stick coatings, in the semiconductor industry for etching silicon chips, in the oil and gas industry for generating hydrogen fluoride, and in the production of uranium for nuclear energy. Additionally, fluorine is used in water fluoridation for dental health and in the production of high-performance materials like fluoropolymers.
Have something to share?
Who is Worksheeto?
At Worksheeto, we are committed to delivering an extensive and varied portfolio of superior quality worksheets, designed to address the educational demands of students, educators, and parents.


























Comments